We aimed to evaluate the performance of the first published software tool1 for the automatic quantification of left ventricular noncompaction (LVNC) based on automatic delineation of the epicardial and endocardial borders of the left ventricular (LV) and trabecular recesses.
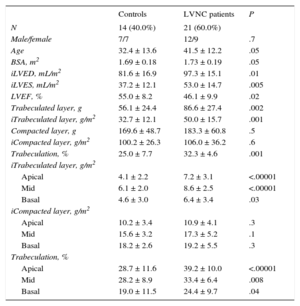
Twenty-one LVNC patients meeting Petersen's criteria2 were compared with 14 control individuals (relatives not meeting LVNC criteria who were free from family mutations). Eleven (52.3%) of the affected patients had systolic dysfunction (8 of those with LV dilatation), 1 had dilatation without systolic impairment, and 1 had LV hypertrophy (20-mm maximum wall thickness). Ten individuals had isolated hypertrabeculation meeting LVNC criteria (Table).
Baseline Characteristics and Summary of Results
| Controls | LVNC patients | P | |
|---|---|---|---|
| N | 14 (40.0%) | 21 (60.0%) | |
| Male/female | 7/7 | 12/9 | .7 |
| Age | 32.4 ± 13.6 | 41.5 ± 12.2 | .05 |
| BSA, m2 | 1.69 ± 0.18 | 1.73 ± 0.19 | .05 |
| iLVED, mL/m2 | 81.6 ± 16.9 | 97.3 ± 15.1 | .01 |
| iLVES, mL/m2 | 37.2 ± 12.1 | 53.0 ± 14.7 | .005 |
| LVEF, % | 55.0 ± 8.2 | 46.1 ± 9.9 | .02 |
| Trabeculated layer, g | 56.1 ± 24.4 | 86.6 ± 27.4 | .002 |
| iTrabeculated layer, g/m2 | 32.7 ± 12.1 | 50.0 ± 15.7 | .001 |
| Compacted layer, g | 169.6 ± 48.7 | 183.3 ± 60.8 | .5 |
| iCompacted layer, g/m2 | 100.2 ± 26.3 | 106.0 ± 36.2 | .6 |
| Trabeculation, % | 25.0 ± 7.7 | 32.3 ± 4.6 | .001 |
| iTrabeculated layer, g/m2 | |||
| Apical | 4.1 ± 2.2 | 7.2 ± 3.1 | <.00001 |
| Mid | 6.1 ± 2.0 | 8.6 ± 2.5 | <.00001 |
| Basal | 4.6 ± 3.0 | 6.4 ± 3.4 | .03 |
| iCompacted layer, g/m2 | |||
| Apical | 10.2 ± 3.4 | 10.9 ± 4.1 | .3 |
| Mid | 15.6 ± 3.2 | 17.3 ± 5.2 | .1 |
| Basal | 18.2 ± 2.6 | 19.2 ± 5.5 | .3 |
| Trabeculation, % | |||
| Apical | 28.7 ± 11.6 | 39.2 ± 10.0 | <.00001 |
| Mid | 28.2 ± 8.9 | 33.4 ± 6.4 | .008 |
| Basal | 19.0 ± 11.5 | 24.4 ± 9.7 | .04 |
BSA, body surface area; i, BSA indexed; iLVED: indexed left ventricular end-diastolic volume; iLVES, indexed left ventricular end systolic volume; LVEF, left ventricular ejection fraction; LVNC, left ventricular noncompaction.
Cardiac magnetic resonance cine images (repetition interval of 2.8ms, echo time of 1.4ms, flip of 60°, matrix of 190 x 200, echo train length of 23, cutting thickness of 8mm, with 30 phases) were reviewed by 2 experienced investigators independently. Fourteen (5.8%) of 242 slices were of insufficient quality. Short axis slices, from the apex to the mitral annulus in end-diastole were analyzed with dedicated software. A standard protocol was used for measurements of LV volumes and wall thickness.
Delineation of the endocardial border, endocardial compacted layer, and pericardial border was performed automatically.1 The trabecular zones are detected inside and around the LV cavity. The software produces measurements of area, volume, and estimates of mass of compacted and noncompacted LV myocardium per slice, and total LV. All measurements are presented as absolute values and are indexed by body surface area. The proportion of trabeculated mass from total LV mass was also calculated. Delineation of the borders was subjectively scored by 2 skilled cardiologists.
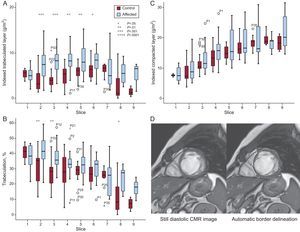
The LVNC patients and control groups showed significant differences in the trabeculated layer mass in most apical and mid slices (slices 2-6) both for absolute and indexed values (Figure). Although the percentage of trabeculation was higher in all slices, it was significant only for apical slices 2 and 3 and basal slice 8. There was no difference in the compacted layer between groups.
A-B: Differences in indexed trabeculated layer and compacted layer per slice in LVNC patients and controls. C-D: Delineation of the endocardial border, endocardial compacted layer and pericardial border was performed automatically by the software. CMR, cardiac magnetic resonance; LVNC, left ventricular noncompaction.
When slices were grouped into apical, mid and basal and all 3 segments showed significantly higher values for absolute trabeculated layer, indexed trabeculated layer and percentage of trabeculation in the LVNC group. As with individual slices, there were no differences in the mass of the compacted layer by segments between groups.
The trabeculated layer and percentage of trabeculation was significantly higher in LVNC patients than in the control group (86.6 ± 27.4g vs 56.1 ± 24.4g; P = .002 and 32.3 ± 4.6% vs 25.0 ± 7.7%; P = .001, respectively).
On multivariate analysis, the indexed trabeculated layer was the variable independently associated with the diagnosis of LVNC (hazard ratio, 1.11; 95% confidence interval, 1.03-1.19; P = .009).
Receiver operating characteristics curve analysis of the 2 variables that differentiated LVNC patients and controls was performed to identify cutoff values. These were 0.82 (95% confidence interval, 0.67–0.96; P = .002) for the indexed trabeculated layer and 0.78 (95% confidence interval, 0.61-0.95; P = .006) for the percentage of trabeculation. A cutoff value of 40.0g/m2 of the indexed trabeculated layer had a sensitivity of 81.0% and a specificity of 78.6%. Similarly, a cutoff value of 27.4% of the percentage of trabeculation had a sensitivity of 90.5% and a specificity of 71.4%.
All (88.6%) but 4 individuals from the total of 35 individuals were appropriately classified. Seventeen (81.0%) of the 21 LVNC patients had values above the 2 cutoffs, and 2 (9.5%) reached only 1 of them.
The performance of the automatic software was evaluated first by the engineers and then by cardiac magnetic resonance expert cardiologists, with very good visual agreement in 96% of the slices. Trabeculation was particularly prominent in apical slices with a 75.6% increase in indexed trabeculated layer in LVNC patients vs the control group, followed by mid and basal segments (41.0% and 39.1%, respectively). The indexed trabeculated layer seems to perform better than the percentage of trabeculation.
Although most definitions of LVNC include a thin, hypokinetic compacted layer in the criteria, we failed to identify any differences, either by slice or globally, in the measurement of compacted myocardium between LVNC patients and controls.
Our automatic software established a higher cutoff of trabeculation (27%) compared with that proposed by Jacquier (20%) using hand delineation of borders.3 With a similar method, Choi et al.4 published a cutoff of 35% in volume trabeculation, when comparing between different groups of cardiomyopathies. Neither of these authors used a trabecula-dedicated software of quantification.
In contrast to the fractal analysis recently published by Captur et al.,5 based on tortuosity measured by pixilation of the line of the endocardial border, our software provides easy to understand clinical measurements of volumes and masses of the compacted and noncompacted LV myocardium. This computationally-assisted method could save valuable diagnostic time compared with traditional processing, thus minimizing the possibility of human error.
In conclusion, we demonstrate the good performance of a new software tool for quantification of hypertrabeculation based on the automatic delineation of borders from cardiac magnetic resonance diastolic images. Cutoffs of normality were established according to trabeculated mass and percentage of trabeculation. Further confirmatory studies are needed.
The investigators are part of a cardiovascular research network of the Carlos III Health Institute (RIC; RD12/0042/0049). This work was supported by the Spanish MINECO, as well as European Commission FEDER funds, under grant TIN2015-66972-C5-3-R.