The benefit of primary angioplasty may be reduced if there are delays to reperfusion. Identification of the variables associated with these delays could improve health care.
MethodsAnalysis of the Codi Infart registry of Catalonia and of the time to angioplasty depending on the place of first medical contact.
ResultsIn 3832 patients analyzed, first medical contact took place in primary care centers in 18% and in hospitals without a catheterization laboratory in 37%. Delays were longer in these 2 groups than in patients attended by the outpatient emergency medical system or by hospitals with a catheterization laboratory (P < .0001, results in median): first medical contact to reperfusion indication time was 42minutes in both (overall 35minutes); first medical contact to artery opening time was 131 and 143minutes, respectively (overall 121minutes); total ischemia time was 230 and 260minutes (overall 215minutes). First medical contact to artery opening time > 120minutes was strongly associated with first medical contact in a center without a catheterization laboratory (OR, 4.96; 95% confidence interval, 4.14-5.93), and other factors such as age, previous coronary surgery, first medical contact during evening hours, nondiagnostic electrocardiogram, and Killip class ≥ III. Mortality at 30 days and 1 year was 5.6% and 8.7% and was independently associated with age, longer delay to angioplasty, Killip class ≥ II, and first medical contact in a center with a catheterization laboratory.
ConclusionsIn more than 50% of patients requiring primary angioplasty, the first medical contact occurs in centers without a catheterization laboratory, which is an important predictor of delay from diagnosis to artery opening.
Keywords
Primary angioplasty is the reperfusion therapy of choice for patients with ST-elevation myocardial infarction (STEMI) when performed within the recommended time frames.1,2 In routine practice, the first medical contact for these patients may involve the outpatient emergency medical services (EMS), hospitals with a catheterization laboratory (HC), hospitals without a catheterization laboratory or regional hospitals (HWC) and primary care centers (PCC). The ideal scenario is the first one, with the patient being transferred directly to the catheterization laboratory after diagnosis, without an intermediate delay at the emergency department. However, the first medical contact often takes place at centers without a catheterization laboratory, resulting in delays to reperfusion.3–8
The infarction code was implemented in June 2009. This protocol consists of a network for treating STEMI using primary angioplasty as the treatment of choice and is coordinated by the public health services provider of Catalonia (CatSalut).9 All patients are systematically included in a registry, which takes into account demographic, procedural and mortality variables and allows results to be analyzed and care to be improved.
The main objective of the study was to determine the times between the onset of pain and artery opening in patients attended through the infarction code, to analyze whether these times differ depending on the place of first medical contact, and to identify the variables predictive of delay to reperfusion. The secondary objective was to determine mortality at 30 days and 1 year, as well as the associated variables.
METHODSOrganization of the Infarction Code ProgramCatalonia is divided into 7 areas (territorial grouping), with 10 HCs which act as referral centers for the assigned area (Figure 1). The infarction code is activated by the center making the diagnosis. If the protocol is activated at an HC, the hospital itself will carry out the interventional procedure. In the other 3 scenarios (PCC, HWC and EMS), the EMS will transfer the patient to the referral hospital to undergo angioplasty, as long as the estimated time is ≤ 90 minutes from the time of first medical contact to arrival at the laboratory. Otherwise, fibrinolysis will be administered, unless contraindicated9.
Study Population and Variables to Be AnalyzedAll patients treated consecutively with primary angioplasty between 2010 and 2011 were analyzed and systematically included in the registry (3832 patients). Four groups were established according to the place of first medical contact: PCC, HWC, HC (which acts as the referral center for the intervention) and EMS. The following time intervals were analyzed: onset of pain-first medical contact, first medical contact-electrocardiogram, first medical contact-activation of the infarction code, first medical contact-arrival at the catheterization laboratory, first medical contact-artery opening (TMO), delay attributable to the angioplasty, and total ischemia time. Analysis of the time intervals was carried out for 3794 patients, as not all the information was available for 38 patients. A distinction was made between daytime first medical contact hours (between 8:00 am and 9:59 pm) and nighttime hours (between 10:00 pm and 7:59 am). The initial electrocardiogram assessment was carried out by the physician in charge of the first medical contact, and the following variables were taken into account in its interpretation: ST-segment elevation, non–ST-segment elevation, presence of left bundle branch block, and nondiagnostic electrocardiogram. The following complications were seen in the acute phase: ventricular tachycardia, ventricular fibrillation, asystole, advanced atrioventricular block, and Killip class. Mortality within the first 24hours, at 30 days, and at 1 year was determined.
Statistical AnalysisContinuous variables are expressed as medians [interquartile range] and categorical variables as absolute frequency. The Student t test was used to compare the means and the chi-square test (or the Fisher exact test, where required) was used to assess the relationship between dichotomous variables. To predict the delay in reperfusion, multivariable logistic regression was used with the dependent variable TMO > 120minutes, and odds ratios (OR) were calculated for the different variables. To predict mortality at 30 days and at 1 year, logistic regression was also used (all variables with a value of P < .1 in the univariable analysis were incorporated in the model); a value of P < .05 was considered to be statistically significant. After the statistical model with the associated variables was obtained, its predictive capacity was analyzed using calculation of the ROC curves and the corresponding area under the curve (an area > 0.8 was considered significant). SPSS 18 software (SPSS, Inc; Chicago, Illinois, USA) was used for the statistical analysis.
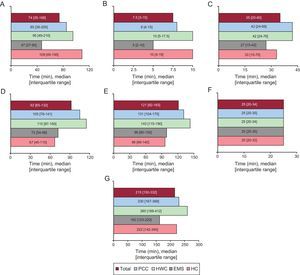
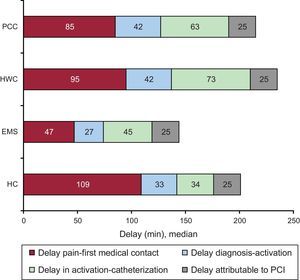
RESULTSBetween 2010 and 2011, primary angioplasty was performed in 3832 patients with STEMI (mean age, 62.6 ± 13.5 years; 79% men). The first medical contact took place in a center without a catheterization laboratory in 2115 patients (55%): 697 in a PCC and 1418 in a HWC. The differences according to the place of first medical contact are shown in Table 1. Patients who were initially attended by the EMS or at an HC had a higher prevalence of previous infarction, history of percutaneous revascularization, and acute-phase heart failure than the other 2 groups. Figure 2A, Figure 2B, Figure 2C, Figure 2D, Figure 2E, Figure 2F, and Figure 2G show the different time intervals according to the place of first medical contact. The PCC and HWC groups showed a longer delay in all times recorded from the onset of pain until artery opening, with the exception of the delay attributable to angioplasty (Figure 3). Artery opening in the PCC and HWC groups was achieved with delays of over 30 and 40minutes from the first medical contact compared with the EMS and HC groups (131 and 143 vs 99 and 96 minutes, respectively; P < .0001) and a TMO ≤ 120 minutes was only achieved in 34% vs 70% of the patients in the EMS groups and 68% in the HC group (P < .0001).
Baseline Characteristics According to Place of First Medical Contact
| Variable | PCC (n = 697) | HWC (n = 1418) | EMS (n = 1141) | HC (n = 576) | Total (n = 3832) | P |
|---|---|---|---|---|---|---|
| Age, y | 61.3 ± 13.7 | 62.8 ± 13.5 | 63 ± 13.3 | 63.5 ± 13.3 | 63 ± 13.5 | .021 |
| Women | 18.7 | 23.1 | 19.5 | 23.3 | 21.3 | .027 |
| Previous MI | 6 | 8.7 | 11.5 | 16 | 10.2 | < .001 |
| Diabetes mellitus | 19.7 | 19.3 | 19.3 | 24 | 20 | .08 |
| Heart surgery | 0.1 | 0.9 | 1.3 | 1 | 0.9 | .08 |
| Previous PCI | 3.6 | 6 | 9.2 | 11.5 | 7.3 | < .001 |
| Suspected thrombosis in stent | 1.9 | 2.3 | 4.1 | 6.4 | 3.4 | < .001 |
| Nondiagnostic ECG | 2.2 | 2.1 | 3.1 | 4.3 | 2.7 | .030 |
| ST-segment elevation | 94 | 93 | 93.4 | 90.5 | 93 | .072 |
| LBBB | 0.3 | 0.8 | 0.7 | 1.2 | 0.8 | .277 |
| Nighttime hours* | 14 | 32.3 | 27.9 | 27.3 | 27 | < .001 |
| Acute-phase complications | 17 | 12 | 26.6 | 20 | 18.5 | < .001 |
| Killip I | 87 | 85.5 | 78.3 | 79 | 82.6 | < .001 |
| Killip II | 7.9 | 7.6 | 9.5 | 8.3 | 8.3 | < .001 |
| Killip III | 0.9 | 2.3 | 3.1 | 4.2 | 6.5 | < .001 |
| Killip IV | 4 | 4.6 | 9.1 | 8.5 | 3.2 | < .001 |
| OTI | 1.6 | 1.6 | 6.1 | 3.5 | 3.2 | < .001 |
| VF | 4.4 | 4.2 | 10.8 | 5.4 | 1 | < .001 |
| VT | 0.4 | 0.8 | 1.8 | 0.9 | 1 | .032 |
| Asystole | 0.3 | 0.5 | 1.7 | 1.7 | 1 | .001 |
| AVB | 2.4 | 3.8 | 6.5 | 5.6 | 4.6 | < .001 |
| Death within 24 hours | 1.4 | 2 | 3.1 | 3.8 | 2.5 | .014 |
| Death within 30 days | 3 | 5.3 | 6.5 | 7.5 | 5.6 | .002 |
| Death within 1 year | 5 | 7.7 | 10.1 | 13 | 8.7 | < .001 |
AVB, advanced atrioventricular block; ECG, electrocardiogram; EMS, emergency medical service; HC, hospital with catheterization laboratory; HWC, hospital without catheterization laboratory; LBBB, left bundle branch block; MI, myocardial infarction; OTI, orotracheal intubation; PCC, primary care center; PCI, percutaneous coronary intervention; VF, ventricular fibrillation; VT, ventricular tachycardia.
Values are expressed as No. (%) or mean ± standard deviation.
Intervals according to the place of first medical contact. A: Time from onset of pain-first medical contact. B: Time from first medical contact-electrocardiogram. C: Time from first medical contact-activation of the infarction code. D: Time from first medical contact-arrival at the catheterization laboratory. E: Time of first medical contact-artery opening. F: Delay attributable to the angioplasty. G: Total ischemia time. EMS, emergency medical service; HC, hospital with catheterization laboratory; HWC, hospital without catheterization laboratory; PCC, primary care center. P < .0001 in all comparisons between groups (with the exception of the delay attributable to angioplasty, P = .589).
Delays from pain onset to coronary reperfusion according to place of first medical contact. EMS, emergency medical service; HC, hospital with catheterization laboratory; HWC, hospital without catheterization laboratory; PCI, percutaneous coronary intervention; PCC, primary care center.
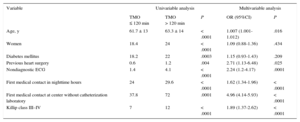
The place of first medical contact (center without a catheterization laboratory, which includes PCCs and HWCs) was the most important predictor of delay to artery opening, together with age, history of coronary revascularization surgery, absence of initial diagnostic electrocardiogram, first medical contact during nighttime hours, and Killip class ≥ III (Table 2). The predictive model obtained showed an optimal capacity for predicting the delay in reperfusion, with an area under the ROC curve of 0.87.
Variables Associated With TMO > 120 Minutes in the Univariable and Multivariable Analyses
| Variable | Univariable analysis | Multivariable analysis | |||
|---|---|---|---|---|---|
| TMO ≤ 120 min | TMO > 120 min | P | OR (95%CI) | P | |
| Age, y | 61.7 ± 13 | 63.3 ± 14 | < .0001 | 1.007 (1.001-1.012) | .016 |
| Women | 18.4 | 24 | < .0001 | 1.09 (0.88-1.36) | .434 |
| Diabetes mellitus | 18.2 | 22 | .0003 | 1.15 (0.93-1.43) | .209 |
| Previous heart surgery | 0.6 | 1.2 | .004 | 2.71 (1.13-6.48) | .025 |
| Nondiagnostic ECG | 1.4 | 4.1 | < .0001 | 2.24 (1.2-4.17) | .0001 |
| First medical contact in nighttime hours | 24 | 29.6 | < .0001 | 1.62 (1.34-1.96) | < .0001 |
| First medical contact at center without catheterization laboratory | 37.8 | 72 | .0001 | 4.96 (4.14-5.93) | < .0001 |
| Killip class III–IV | 7 | 12 | < .0001 | 1.89 (1.37-2.62) | < .0001 |
ECG, electrocardiogram; 95%CI, 95% confidence interval; OR, odds ratio; TMO, time from first medical contact-artery opening.
Values are expressed as percentage or mean ± standard deviation.
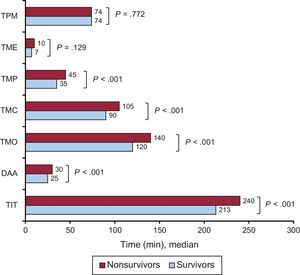
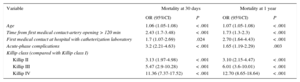
Total mortality was 5.6% at 30 days after the infarction and 8.7% at 1 year. This was significantly higher according to the Killip class (P < .001): Killip I, 2.2% at 30 days and 4.3% at 1 year; Killip II, 10% and 16%; Killip III, 16.5% and 30%, and Killip IV, 38% and 46%. The time intervals were significantly longer in nonsurvivors than in survivors (Figure 4). The variables related to mortality at 30 days and 1 year after the infarction were age, TMO > 120 minutes, presence of acute-phase complications, Killip class ≥ II and the first medical contact at an HC (Table 3). The resulting predictive model showed an optimal capacity for predicting mortality at 30 days (area under ROC curve 0.88) and at 1 year (area under ROC curve 0.85).
Differences in the time intervals (in medians) between survivors and nonsurvivors at 1 year. DAA, delay attributable to angioplasty; TIT, total ischemia time; TME, time from first medical contact-electrocardiogram; TMC, time from first medical contact-arrival at the catheterization laboratory; TMO, time from first medical contact-artery opening; TMP, time from first medical contact-activation of the infarction code; TPM, time from onset of pain-first medical contact.
Variables Associated With Total Mortality at 30 Days and 1 Year in the Multivariable Analysis
| Variable | Mortality at 30 days | Mortality at 1 year | ||
|---|---|---|---|---|
| OR (95%CI) | P | OR (95%CI) | P | |
| Age | 1.06 (1.05-1.08) | < .001 | 1.07 (1.05-1.08) | < .001 |
| Time from first medical contact-artery opening > 120 min | 2.43 (1.7-3.48) | < .001 | 1.73 (1.3-2.3) | < .001 |
| First medical contact at hospital with catheterization laboratory | 1.7 (1.07-2.69) | .024 | 2.70 (1.64-4.43) | < .001 |
| Acute-phase complications | 3.2 (2.21-4.63) | < .001 | 1.65 (1.19-2.29) | .003 |
| Killip class (compared with Killip class I) | ||||
| Killip II | 3.13 (1.97-4.98) | < .001 | 3.10 (2.15-4.47) | < .001 |
| Killip III | 5.47 (2.9-10.28) | < .001 | 6.01 (3.6-10.01) | < .001 |
| Killip IV | 11.36 (7.37-17.52) | < .001 | 12.70 (8.65-18.64) | < .001 |
95%CI, 95% confidence interval; OR, odds ratio.
The main findings of this study are as follows: a) more than half of the patients who underwent primary angioplasty in this environment were initially seen at centers without a catheterization laboratory; b) time intervals differed significantly according to the place of first medical contact, with longer delays for the patients initially seen at PCCs and regional hospitals; c) the place of first medical contact acted as an independent predictor of delay to reperfusion, and d) delay to reperfusion acted as an independent predictor of mortality at 30 days and 1 year after infarction in the patients in this series.
Relationship Between the Place of First Medical Contact and Delays to ReperfusionIn this series of consecutive patients treated with primary angioplasty in the first 2 years of the implementation of the infarction code, in over half of the patients, the first medical contact took place at a center without a catheterization laboratory, including regional hospitals (67%) and PCCs (33%). This group recorded the longest delays to reperfusion and a TMO ≤ 120minutes was only achieved in one third of the patients compared with 70% and 68% of the EMS and HC groups. Other contemporary registries show similar results.4,7,8 The implementation of networks to improve reperfusion therapy in STEMI has significantly increased the number of primary angioplasties.10 However, these protocols must take into account the possibility of excessive delays occurring in their application, which limit the benefits. Another relevant objective is therefore that the activation times do not differ according to the place of first medical contact. Both clinical practice guidelines1 and the infarction code protocol9 consider the ideal scenario to be one in which the EMS is the first medical contact in these patients, and our series provides data that confirm this indication. Nevertheless, this was only the case for 3 of every 10 patients in our study, and this percentage is repeated in the literature.4,7
These results can be explained by various factors. First, there is a delay between the onset of pain and the request for medical care, which has an impact on total ischemia time. In addition, the place of first medical contact may vary according to the patient's knowledge of their condition, symptom severity, and the influence of demographic and cultural factors.11 If there is a lack of awareness of severity, the patient is likely to go to the nearest center (probably a regional hospital or PCC), while a patient with a more severe condition or one who is aware of their condition may initially seek emergency care from the EMS12 or go directly to the hospital where they had previously been treated.
After requesting medical care, the minutes that elapse from the diagnosis of infarction until the indication of angioplasty are key to starting the transfer to the catheterization laboratory and this time can be reduced with a rapid initial selection. Miedema et al.2 reported that in 64% of patients the factors related to the delay to reperfusion originated in the place of first medical contact (eg, errors in recognizing the symptoms and the initial interpretation of the electrocardiogram, waiting time for the patient to be transferred). Finally, the times required to reach the catheterization laboratory and for artery opening have their own constant factors,2 such as geography and climate in one case (in the event of transfer between centers) and the duration of the interventional procedure in the other. However, the total time to reperfusion can be reduced if diagnosis and activation are performed promptly.13
Predictive Variables of Time From First Medical Contact-artery Opening > 120MinutesThe variables associated with a TMO > 120 minutes were age, history of coronary revascularization surgery, having a nondiagnostic initial electrocardiogram, first medical contact during nighttime hours (between 10:00 pm and 7:59 am), place of first medical contact (center without a catheterization laboratory vs EMS and HC), and Killip class ≥ III.
Older patients may have more atypical initial symptoms,14 which leads them to seek medical care at a later stage and delays the diagnosis. They also have a higher incidence of acute-phase complications,15 which contributes to the delay to reperfusion while they are stabilized.
Patients who have a nondiagnostic initial electrocardiogram have a 2-fold risk of delay. This relationship is obvious, and while the electrocardiogram criteria for indication of reperfusion therapy are well defined,1 it is not uncommon to find cases with borderline criteria in routine practice,16 or cases that are not evident until hours later, when the infarction has progressed. Nevertheless, it has been demonstrated that sensitivity can be increased in these cases by performing serial electrocardiograms in the 30minutes that follow.17 In addition, patients with a history of heart surgery usually have more comorbidities,18,19 a higher incidence of complications19 and less ST-segment elevation in the acute phase,18 which delays diagnosis and artery opening.
The first medical contact during nighttime hours increased the risk of a delay in the indication of angioplasty by over 50%. This relationship has been reported in the literature, although it varies according to the definition of nighttime hours.5,7,8 Healthcare staffing levels during this time period are usually lower than during the day in most centers, which can obviously contribute to slower diagnosis and transfer to the catheterization laboratory.
The place of first medical contact is a highly important predictor and, as previously mentioned, the ideal scenario for minimizing delay times is for the EMS to establish the diagnosis and indication of primary angioplasty.
Finally, patients with heart failure or other complications need to be stabilized prior to transfer, which can involve invasive mechanical ventilation support, perfusion of vasoactive amines and, in some patients, placement of a provisional pacemaker; all of this obviously results in a delay to reperfusion5.
Factors Related to Mortality at 30 Days and 1 YearThe variables related to mortality at 30 days and 1 year were age, longer time to artery opening (TMO > 120 minutes), presence of acute-phase complications, Killip class ≥ II, and first medical contact at an HC.
Both age and the degree of heart failure are variables traditionally associated with worse prognosis.15 With regard to TMO, there is a strong relationship between the delay to artery opening after the infarction, which is independent of the other associated risk variables, and is progressive: the longer the delay, the higher the mortality.4,20
Finally, an important finding was the higher mortality rate in patients whose first medical contact took place at an HC, despite being one of the groups with the fewest delays to artery opening. The variables that may help explain this difference are a higher frequency of Killip I and a lower incidence of complications in the PCC and HWC groups compared with the HC group. Even so, these results must be interpreted with caution, as there may be a residual confounding bias attributable to other variables not included in the registry.
Proposals for Improvement and Implementation of Health Care QualityThe results of this study allow several proposals to be made for improving health care quality. The delays with the strongest impact on total ischemia time were those taking place between the onset of pain and the request for medical care, between diagnosis and activation of the infarction code, and between activation of the code and arrival at the catheterization laboratory, while the time taken for the interventional procedure was similar among the 4 groups.
Since the implementation of the infarction code, educational campaigns have been carried out for the population and a telephone number is available (112) in the event of a suspected infarction, with the aim of making the EMS the place of first medical contact. Courses are also regularly imparted on updates in the treatment of infarction, aimed at health care professionals involved in the first medical care of these patients, as well as reviews of the protocols according to the scientific evidence available and the particular nature of each center. In addition, it is important to carry out regular analyses of the time intervals (both in general and in each center involved in the first medical contact) to identify the factors that have the greatest impact on unnecessary delays to artery opening.21 The latter is of the utmost importance in centers without a catheterization laboratory (from the first contact with the patient until the start of the transfer), given its relevance in the scenario of the initial STEMI treatment.
Finally, it is important to be aware of patients with a higher risk of delay in diagnosis (eg, older patients, those with a history of heart surgery), so as not to underestimate them.
LimitationsOur study has some limitations. On the one hand, given that the analysis was performed in a single autonomous community (Catalonia), the results may not be applicable to the rest of the communities, which have different health care organizations and geographies. That said, the results are quite similar to those of other contemporary registries4,7,8 and ours is a large series of patients included systematically, which makes the results more reliable. On the other hand, due to limitations in the registry itself, some variables have not been recorded, such as the time between the first medical contact and the start of the transfer to the HC for patients in the PCC and HWC groups, or the distance in kilometers to the HC in the case of transfers between centers. Nevertheless, the results obtained reasonably describe the operation of the infarction code program in its first 2 years of implementation, and variables have been identified which significantly influence its operation, with a high predictive value. Finally, given that it is a registry and not a randomized study, there may be a confounding bias attributable to other variables not included in the registry. Nevertheless, the registry contains a large series of consecutive patients, and is therefore a reliable reflection of routine clinical practice.
CONCLUSIONSThe first medical contact for patients with STEMI in our environment, with a network organized to perform primary angioplasty as the treatment of choice, takes place at centers without a catheterization laboratory in more than half of the cases. This leads to patients being transferred to another hospital, which involves longer delays to artery opening. The place of first medical contact is an important, independent predictor of delay to reperfusion, together with age or a lack of an initial diagnostic electrocardiogram, and other lesser known factors such as the time of day of the first medical contact, history of heart surgery, and the presence of heart failure (Killip class ≥ III). Identifying these variables is of the utmost importance for improving the future care of these patients. Finally, the delay to artery opening is an independent predictor of mortality at 30 days and 1 year.
- –
The implementation of networks for the treatment of STEMI can help reduce the delay from diagnosis to artery opening through primary angioplasty. This is a highly important factor, as delay to reperfusion has an impact on prognosis and can minimize the treatment benefit.
- –
The ideal scenario is for the first medical contact to take place at the EMS, and for the patient to be directly transferred to the catheterization laboratory, avoiding the delays which would occur when initially visiting a center without a catheterization laboratory.
- –
In an environment with a STEMI network, more than half of patients who undergo primary angioplasty have their first medical contact at centers without a catheterization laboratory.
- –
The place of first medical contact is an important, independent predictor of delay to reperfusion, together with age or a lack of initial diagnostic electrocardiogram, and other lesser known factors such as the time of day of the first medical contact, history of heart surgery, and the presence of heart failure.
- –
Delay to reperfusion is an independent predictor of mortality at 30 days and 1 year.
Assistance was provided for conducting the statistical analysis of the study, funded by the Codi Infart program of the Plà director de malalties de l’aparell circulatori.
CONFLICTS OF INTERESTNone declared.
To all the professionals involved in the Codi Infart program, who, through their effort and dedication, are able to continuously improve the care of these patients. To Josep Ramon Marsal, for his assistance with the statistical analysis.