The aim of this study was to assess patterns of treatment persistence in a cohort of male Spanish workers receiving statin therapy for primary prevention of cardiovascular disease.
MethodsThis descriptive study was conducted within the framework of the prospective longitudinal Aragon Workers’ Health Study (N = 5400). Incident male statin users were identified based on data collected from the regional government's medication consumption information system. Patterns of treatment persistence with statins prescribed for primary cardiovascular disease prevention were assessed and the relevance of potential predictors explored.
ResultsAmong the 725 new statin users, less than one third remained persistent during the 1 year of follow-up. About 15% of nonpersistent users discontinued statin therapy after dispensation of the first prescription; of these, 42.1% did not recommence treatment within the following year. Factors reducing the likelihood of treatment discontinuation were older age (HR, 0.55; 95%CI, 0.39-0.77) and cotreatment with antihypertensive drugs (HR, 0.68; 95%CI, 0.56-0.82). No association was observed between treatment persistence and cotreatment with antidiabetic or antithrombotic drugs, baseline low-density lipoprotein levels, or total cholesterol levels. However, persistence was influenced by the type of statin first prescribed.
ConclusionsOur analysis of a cohort of healthy male workers revealed poor statin persistence. These findings underscore the need for a better understanding of patterns of statin use, especially in apparently healthy individuals, and for the incorporation of patient behavior into prescribing decisions.
Keywords
Statins, by reducing blood cholesterol levels, decrease cardiovascular (CV) events and all-cause mortality in individuals with and without evidence of CV disease.1 While rates of statin use are high in most European populations,2 cholesterol control in these same populations is generally poor,3,4 indicating that statin effectiveness may not correspond exclusively to the efficacy demonstrated in clinical trials, but rather is affected by real-life patterns of statin use. Among the contextual characteristics and behaviors that influence goal attainment, a key factor is “patient persistence” (i.e., whether the patient persists with treatment).4–7 Indeed, longer duration statin therapy is associated with improved clinical outcomes.4–6
Several studies have assessed rates of treatment persistence with statins for secondary prevention of CV disease.5,6,8 However, less is known about treatment persistence in the case of statin use for primary prevention of CV disease in young people. Both primary prevention regimens and young age are associated with poorer treatment adherence,9,10 probably because the users in question do not perceive themselves as being “sick”. Describing patterns of statin treatment persistence in this type of population is essential to enhance persistence and ultimately improve the prevention of chronic conditions.
The objective of this study was to assess the pattern of persistence with statin therapy for primary prevention of CV disease in a cohort of male Spanish workers.
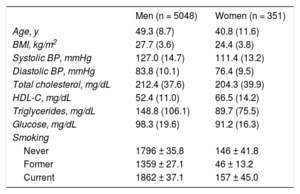
METHODSStudy Population and Data SourcesThe Aragon Workers’ Health Study (AWHS) is a prospective longitudinal study designed to characterize cardiometabolic factors and assess subclinical atherosclerosis in a middle-aged Mediterranean working population. For that purpose, we analyzed data from routine annual health examinations of workers at a Spanish automobile assembly plant in Figueruelas, Zaragoza (Spain), who voluntarily agreed to participate in the study. The cohort was recruited between February 2009 and May 2010, and consisted of 5400 workers. Participants aged 40 to 55 years were additionally invited to participate in triennial imaging examinations to assess the presence of subclinical atherosclerosis and complete additional questionnaires of CV and lifestyle factors. At baseline, 587 (94.5% men) were selected for this intensive follow-up group. Most of the workers witihin the cohort were involved in manual jobs (86.5% of men and 60.5% of women). Other baseline characteristics are shown in Table 1.11 The cohort showed a high prevalence of CV risk factors and subclinical atherosclerosis but a low prevalence of clinical CV disease. Active follow-up of participants is expected to continue through 2020. Further information on the AWHS can be found in Casasnovas et al.11
Aragon’ Workers Health Study Baseline Characteristics
| Men (n = 5048) | Women (n = 351) | |
|---|---|---|
| Age, y | 49.3 (8.7) | 40.8 (11.6) |
| BMI, kg/m2 | 27.7 (3.6) | 24.4 (3.8) |
| Systolic BP, mmHg | 127.0 (14.7) | 111.4 (13.2) |
| Diastolic BP, mmHg | 83.8 (10.1) | 76.4 (9.5) |
| Total cholesterol, mg/dL | 212.4 (37.6) | 204.3 (39.9) |
| HDL-C, mg/dL | 52.4 (11.0) | 66.5 (14.2) |
| Triglycerides, mg/dL | 148.8 (106.1) | 89.7 (75.5) |
| Glucose, mg/dL | 98.3 (19.6) | 91.2 (16.3) |
| Smoking | ||
| Never | 1796 ± 35.8 | 146 ± 41.8 |
| Former | 1359 ± 27.1 | 46 ± 13.2 |
| Current | 1862 ± 37.1 | 157 ± 45.0 |
BMI, body mass index; BP, blood presure; HDL-C, high-density lipoprotein cholesterol
Data are expressed as mean ± standard deviation or no. (%).
Reproduced with permission from Casasnovas et al.11
We designed the present observational descriptive study to assess persistence with statin therapy in the AWHS cohort. Statin prescription data corresponding to AWHS participants was gathered from Farmasalud, the government medication consumption information system for Aragon, which is the Spanish autonomous community where the automobile factory is located. This database collects data on all prescriptions dispensed at pharmacies in Aragon through the public health care system (i.e., prescribed by either a company physician or a general practitioner from the public health care system). Prescriptions issued by private physicians, insurance companies, or in-hospital consumption, are not collected in Farmasalud. Each record in the data source corresponds to a prescription and contains the following information: an anonymous patient code, patient sex and birth date, dispensing date, Anatomical Therapeutic Chemical code of the prescribed drug, number of defined daily dose, and number of packages dispensed. Drugs were classified according to the 2015 version of the World Health Organization Anatomical Therapeutic Chemical code/Defined Daily Dose System.12 Via an encrypted code provided by AWHS researchers, we identified prescriptions for statins, both alone (Anatomical Therapeutic Chemical code C10AA) and in combination with other lipid-modifying agents (C10BA), issued between January 1, 2010 and December 31, 2014.
From the AWHS database we gathered data on CV events experienced by AWHS participants (date and event type), as well as levels of total cholesterol and other lipid fractions recorded at annual medical examinations. The low-density lipoprotein cholesterol (LDL-C) value for each patient and examination was calculated using the Friedewald formula,13 based on total cholesterol, high-density lipoprotein cholesterol, and triglyceride values.
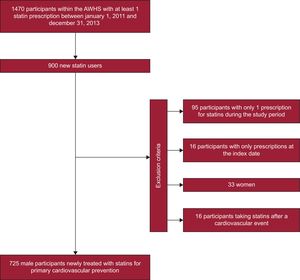
By patient, the date of the first statin dispensing was defined as the index date. Analyses were restricted to newly-treated statin users, defined as those who had not received any statin prescription during the 12 months preceding the index date, between January 1, 2011 and December 31, 2013. Due to their low frequency in the AWHS population, women (4.2%) and men who had experienced a CV event before beginning statin therapy (secondary prevention [3.9%]) were excluded from our analyses.
Assessment of PersistencePersistence was defined as continuation of treatment during a follow-up period of 1 year from the index date. Persistence was determined by measuring the time (gap) between a dispensation of a drug and the next dispensation. For this reason, participants who received only 1 statin prescription during the study period or who received 1 or more prescriptions only at the index date were excluded from the analysis. Participants were considered nonpersistent if the gap between refills was over twice the duration of the preceding prescription. While other authors have defined this gap by using a fixed number of days, we selected an alternative approach given the variable time window (1-2 months) between dispensations in the autonomous community of Aragon. The number of days of medication supplied was estimated on the basis of the number of pills and packages. In Spain, most statins are sold in packages of 28 pills, with the exception of some presentations of lovastatin, pravastatin/fenofibrate, and atorvastatin/ezetimibe, which are sold in packages of 30 pills. Of all the prescriptions dispensed to AWHS participants, only 2 corresponded to lovastatin. The usual prescribed statin dosage is 1 pill per day. Therefore, assuming that each package contained 28 daily doses, the maximum allowable gap was 56 days. The accumulation of supplies over time was not considered (i.e., only the number of pills from the most recently dispensed prescription was used to evaluate the gap). Participants were censored if the gap allowed was exceeded without purchasing a new prescription or upon reaching the end of the study period (if they had been persistent throughout the follow-up period). Nonpersistent participants were categorized as users who restarted statin therapy after a period of discontinuation, or users who simply discontinued treatment (and received no corresponding statin prescription after the end of the allowable maximum gap).
Statistical AnalysisThe characteristics of newly-treated statin users were recorded. Age at the index date was categorized as follows: < 50 years, 50 to 54 years, 55 to 59 years, and ≥ 60 years. Concomitant antidiabetic, antihypertensive or antithrombotic therapy were assessed by taking into account the existence of any prescription for each process, respectively, during the period between the statin index date and the discontinuation date or end of follow-up. Baseline total cholesterol and LDL-C values, recorded at the patient's last medical examination before the index date, were categorized as follows: < 100, 100 to 154, 155 to 189 and ≥ 190mg/dL for LDL-C; and < 200, 200 to 240, and > 240mg/dL for total cholesterol. These categories were established in line with the reference values provided in the European guidelines for CV disease prevention14 for individuals with low/moderate CV risk, such as those in our study population.
Persistence rates were longitudinally analyzed using the Kaplan-Meier method. Statistical differences between curves were assessed using the log-rank test. The likelihood of nonpersistence over the follow-up period was determined by multivariate analysis with Cox regression. Hazard ratios (HR) and 95% confidence intervals (95%CI) were calculated, adjusting for age group at the index date, concomitant antidiabetic, antihypertensive or antithrombotic therapy, baseline LDL-C level, and baseline total cholesterol level.
A sensitivity analysis was performed to account for the possible influence of the selected interval duration on participant classification. The proportion of persistent statin users was recalculated by applying interval durations of 1.5 and 2.5 times the number of daily doses (i.e., 42 and 70 days, respectively).
All analyses were performed using STATA version 12.1 (StataCorp, College Station, Texas, United States).
EthicsParticipants in the AWHS provided written informed consent when became part of it, and the present study was approved by the Clinical Research Ethics Committee of Aragon.
RESULTSCharacteristics of New Statin UsersAmong the 5400 individuals in the AWHS, 725 new statin users fulfilled the inclusion criteria (Figure 1), with a mean (standard deviation) age of 54.7 (5.1) years. Categorization by age revealed the following breakdown: < 50 years, 13.1%; 50 to 54 years, 31.9%; 55 to 59 years, 36.8%; ≥ 60 years, 18.2%.
The median (range) baseline LDL-C value was 159.6 (58.0–294.8) mg/dL. The distribution of statin users in the different LDL-C categories was: < 100mg/dL, 26 (3.8%); 100 to 154mg/dL, 273 (39.7%); 155 to 189mg/dL, 294 (42.8%); and ≥ 190mg/dL, 94 (13.7%). The median (range) total cholesterol value was 247.0 (142.0–481.0) mg/dL, and the distribution of statin users was: < 200mg/dL, 85 (11.8%); 200-240mg/dL, 213 (29.5%); > 240mg/dL, 425 (58.8%). The median (range) of other CV risk factors was: glucose level of 98.0 (61.0–292.0) mg/dL; diastolic blood pressure of 86.0mmHg (60.0–129.0), systolic blood pressure of 128.0mmHg (90.0–220.0), and body mass index of 27.8kg/m2 (19.3–43.6).
During the corresponding follow-up period, 49.8% of statin users were being cotreated with other drugs for CV disease prevention: 36.3% with 1 additional drug, 11.7% with 2 additional drugs, and 1.8% with 3 additional drugs. Specifically, 41.4% were taking an antihypertensive, 12.3% an antithrombotic, and 11.5% an antidiabetic drug.
The most frequently prescribed statin at the index date was simvastatin (40.4% of new users), followed by atorvastatin (24.7%), rosuvastatin (24.4%), pitavastatin (5.8%), pravastatin (2.3%), simvastatin/ezetimibe (1.4%), fluvastatin (0.8%), and lovastatin (0.1%).
The 22.6% of statin prescriptions were issued by a company doctor and 77.4% by a general practitioner from the public health care system.
Assessment of PersistenceOf the 725 newly-treated statin users, 29.5% continued treatment for the duration of the 1-year follow-up period. The average period between the index date and treatment discontinuation was 211.3 days. According to the Kaplan-Meier analysis, half of all new statin users were treatment-persistent at 184 days after the index date, and 36.9% at 270 days after the index date. The survival curve dropped steeply at 56 days, the end of the first allowable gap (28 daily doses x 2), indicating that about 15% of new statin users discontinued treatment right after the first prescription. Among those that discontinued treatment, 57.9% restarted a statin therapy later, while 42.1% were not dispensed any other statin during the follow-up period.
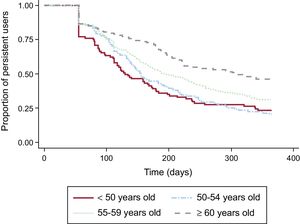
Age-linked differences in persistence rates were observed (log-rank, P < .0001) (Figure 2), with persistence increasing with age. Analysis of the corresponding persistence rates for the 3 most commonly prescribed statins revealed higher rates for new simvastatin users than for rosuvastatin or atorvastatin users (log-rank P < .05).
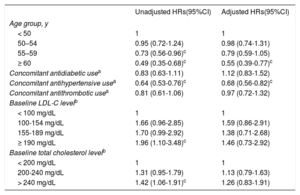
Factors Associated With PersistenceIn the bivariate analysis, the crude HR for discontinuation of statin prescription (Table 2) was significantly lower in the 2 highest age groups (55–59 years and ≥ 60 years) than in the youngest age group (< 50 years), and in users of concomitant antihypertensive treatments than in those not taking concomitant treatments for the prevention of CV disease. By contrast, higher HR values were observed for participants with baseline total cholesterol levels > 240mg/dL compared with those with levels < 200mg/dL, and in participants with baseline LDL-C levels ≥ 190mg/dL compared with those with levels < 100mg/dL. Multivariate analysis (Table 2) revealed a statistically significant HR only for statin users in the highest age group (≥ 60 years old) (HR, 0.55; 95%CI, 0.39–0.77) and in those cotreated with antihypertensive drugs (HR, 0.68; 95%CI, 0.56–0.82).
Hazard Ratios for Discontinuation of Statin Therapy in a Cohort of Male Spanish Workers With no Prior Cardiovascular Events. Statin Therapy Began Between January 1, 2011 and December 31, 2013
| Unadjusted HRs(95%CI) | Adjusted HRs(95%CI) | |
|---|---|---|
| Age group, y | ||
| < 50 | 1 | 1 |
| 50–54 | 0.95 (0.72-1.24) | 0.98 (0.74-1.31) |
| 55–59 | 0.73 (0.56-0.96)c | 0.79 (0.59-1.05) |
| ≥ 60 | 0.49 (0.35-0.68)c | 0.55 (0.39-0.77)c |
| Concomitant antidiabetic usea | 0.83 (0.63-1.11) | 1.12 (0.83-1.52) |
| Concomitant antihypertensive usea | 0.64 (0.53-0.76)c | 0.68 (0.56-0.82)c |
| Concomitant antithrombotic usea | 0.81 (0.61-1.06) | 0.97 (0.72-1.32) |
| Baseline LDL-C levelb | ||
| < 100 mg/dL | 1 | 1 |
| 100-154 mg/dL | 1.66 (0.96-2.85) | 1.59 (0.86-2.91) |
| 155-189 mg/dL | 1.70 (0.99-2.92) | 1.38 (0.71-2.68) |
| ≥ 190 mg/dL | 1.96 (1.10-3.48)c | 1.46 (0.73-2.92) |
| Baseline total cholesterol levelb | ||
| < 200 mg/dL | 1 | 1 |
| 200-240 mg/dL | 1.31 (0.95-1.79) | 1.13 (0.79-1.63) |
| > 240 mg/dL | 1.42 (1.06-1.91)c | 1.26 (0.83-1.91) |
95%CI, 95% confidence interval; HR, hazard ratio; LDL-C, low-density lipoprotein cholesterol.
N = 725 in all analyses, except analysis of LDL-C levels. In those cases (i.e., bivariate analysis of LDL-C and the multivariate regression model), 687 participants were included.
The proportion of new statin users considered treatment-persistent was 13.2% and 42.8%, applying gaps of 1.5 and 2.5 times the number of daily doses dispensed, respectively. Application of these distinct gap durations confirmed the results of the Cox regression analysis (data not shown).
DISCUSSIONOur analysis of a cohort of male workers revealed that less than one third of new statin users remained persistent with treatment for the duration of the first year of therapy. The likelihood of remaining persistent increased with age. A previous meta-analysis of studies evaluating predictors of nonadherence to statins15 found a lower likelihood of persistence and adherence among the youngest (< 50 years) and oldest (> 70 years) statin users. While our findings confirmed the association between poor treatment persistence and younger age, we were unable to evaluate the pattern in those older than 70 years, who were not represented in the AWHS cohort.
Half of the new statin users in the study population were taking at least 1 other treatment for the prevention of CV disease, with 41.4% receiving concurrent antihypertensive therapy. The likelihood of persistence was higher for these users, in line with previous reports.16,17 However, those same studies also identified cotreatments for other CV risk factors, such as antidiabetic or antithrombotic drugs, as predictors of statin treatment continuity. In addition to the recognized influence of factors such as patient age or socioeconomic position,16,17 the presence of other CV risk factors may influence long-term treatment in individuals free of CV disease, probably given the greater perceived risk of heart disease in these patients. According to some authors, this association does not apply to the oldest adults, in whom statin adherence is also influenced by other factors such as regimen complexity, the increased incidence of adverse events, and impaired cognition.15 Moreover, it has been hypothesized that individuals that have been previously warned by their physician of the risks of high blood cholesterol levels are more aware of the importance of CV disease control and thus may be more treatment adherent.8 To our knowledge, only 1 previous study, conducted in a Swedish population8, analyzed the possible association between baseline cholesterol values and persistence. The authors reported that high baseline LDL-C values were significantly associated with a lower discontinuation rate in nondiabetic patients with no previous CV events. By contrast, we observed no clear association between baseline cholesterol values and treatment persistence after adjusting for other variables.
Simvastatin was the most commonly prescribed statin in our study cohort (40.4% of new statin users). According to current European recommendations,14 the type of statin prescribed should be selected on the basis of the patient's total CV risk and on the expected percentage reduction in LDL-C levels, which is dependent on the baseline value. The frequent prescription of a low-potency statin as an index drug seems reasonable, given that workers in the study cohort were mostly healthy, with median LDL-C and total cholesterol values defined as moderate-high and less than 15% presenting 2 or more concomitant CV risk factors. Our results also showed that simvastatin users were significantly more likely to remain treatment-persistent than individuals receiving higher-potency statins such as atorvastatin and rosuvastatin. Similarly, in their study of a middle-aged population free of CV disease in Quebec, Canada, Perreault et al.16 reported an increased risk of nonpersistence in patients prescribed atorvastatin compared with those prescribed simvastatin or pravastatin. Conversely, a large population-based study of users of statins for primary CV disease prevention,17 conducted in the United Kingdom, found no association between the type of statin prescribed and the risk of discontinuation. It is difficult to reach an overall conclusion based on the results of multiple studies of predictors of persistence due to the variability of the populations studied, the data available, and the methods used. The exclusion of women from our analysis limits the comparability of our findings, particularly given the demonstrated association between sex and adherence and persistence.9,15–17 Furthermore, factory work is likely associated with several social, medical, and health care utilization characteristics that influence patient behavior15 in ways that may differ from those in the general population.
We found that about 15% of new statin users discontinued treatment immediately after purchasing the first prescription. It is estimated that almost 12% of statin users experience adverse effects attributed to these drugs,7 which may explain why these patients discontinued treatment early and refused to restart it. Indeed, our results show that 42.1% of all users classified as nonpersistent did not recommence statin therapy during the 1-year follow-up period. This finding is in agreement with the results of the aforementioned United Kingdom population study,17 in which 45% of primary prevention statin users discontinued treatment and did not recommence within the following year. The authors of that study reported that female sex, older age, lower body mass index, and dementia were associated with an increased risk of discontinuation and a lower likelihood of restarting statin treatment.
LimitationsThe present study has several limitations. The main ones pertain to the data source used. Because the Farmasalud database only records prescriptions issued via the public health system, persistence may have been misestimated in some cases. However, it is unlikely that participants would have switched from a public health care physician to a private physician. Participants in the Aragon Workers’ Health Study have easy access to a physician, whose office is located close to their work area. Accordingly, workers usually visit the same physician in the company. Alternatively, they may visit a general practitioner within the publicly-funded health care system, in which case the prescription data would have been recorded in the same data source.
Also absent from our data source were the prescribed dose and the quantity of drug dispensed (only the number of defined daily dose was available). Given the recognized limitations of defined daily dose as a unit of measurement,18 we converted the number of pills to the daily dose based on the commercialized presentations of statins. In our opinion, this approach is valid when the recommended dose always equals a fixed number of pills.
Our data source provided information on a limited number of variables pertaining to the study participants and their treatment regimens. Information on the reasons to start treatment, family history, clinical characteristics, prescriptions for treatments other than those for the prevention of CV disease, physician characteristics, or patient attitude was not available, and would have been of interest for the identification of additional predictors of treatment persistence. Certainly, the lack of information related to the reasons to prescribe poses a determining limitation for the validity of the study, and is mainly due to the fact that the data source was not designed for this purpose.
A common limitation of analyses of drug-dispensing data is the assumption that drug purchase equates to drug consumption. Our objective was to determine rates of persistence, which may be a more appropriate indicator of treatment adherence than adherence, given that it is based on the continuity of prescription refilling behavior rather than on the number of doses available. The fact that an individual regularly attends the pharmacy to purchase their medication is a probable indicator that he is taking the prescribed medication.
In addition to the aforementioned limitations related to the data source, the variety of methodologies used to define and assess treatment persistence, as well as the lack of standardized approaches, complicates the calculation and comparison of results. Considerable variability is observed, for example, in the maximum allowable duration between dispensations. Two respective studies of Swedish8 and British17 populations measured treatment persistence in new statin users, applying a gap of 90 days from the date of the last prescription refill. The authors of the Swedish study based this decision on national regulations, which can result in a time window of up to 3 months between refills. As expected, both studies reported higher rates of statin treatment persistence (68% and 53%, respectively) than that reported here (29.5%). Although we did not initially consider a gap of 2.5 times the daily dose as the most appropriate value for evaluation of treatment persistence, the proportion of persistent users obtained using this value (42.8% of new statin users) appeared more realistic and was more in line with findings in the other populations. While our sensitivity analyses supported the robustness of the results and the gap durations applied, it should be noted that clearly defined methods and criteria for the calculation of persistence indicators are essential to ensure comparability. Several common scenarios can contribute to underestimation of statin treatment persistence. For instance, some participants can be misclassified as nonpersistent when their treatment is switched from a statin to a non-statin lipid-lowering drug. This situation is unlikely however, given that statins, alone or in combination, are recommended as the first line of treatment for hypercholesterolemia.14 Similarly, a user could purchase their first package of statins after the medical consultation but wait several days before starting treatment, thus exceeding the permitted gap and resulting in misclassification as a discontinuer.
Finally, our study population consisted only of healthy and relatively young men, which complicates extrapolation of our results to the general population of new statin users. The study population corresponds to a cohort of volunteers who are particularly aware of CV risk and undergo annual reviews and clinical analyses, as well as receiving regular and detailed health advice. We suspect that persistence with statin treatment for primary prevention of CV diseases in health care settings distinct from the AWHS may be even lower.
Nonetheless, our findings constitute an important contribution to knowledge about patterns of medication use for the prevention of CV disease, including statins. The prescription of statins in populations such as that described here remains controversial. These drugs are recommended in current clinical guidelines14 as the first-line treatment for high CV risk patients with hypercholesterolemia (nonfamilial) or combined hyperlipidemia, and are widely accessible due to their low cost. However, evidence indicates that high-potency statins are associated with an increased risk of adverse effects, including new-onset diabetes, particularly in patients at risk of developing diabetes.19 Furthermore, some authors20 have reported that pharmaceutical industry-sponsored economic evaluations of these drugs, particularly statins used for primary prevention of CV disease, generally favor the cost-effectiveness profile of the products in question.
Nevertheless, the importance of patient behavior in the management of CV risk factors is undeniable. A systematic review6 of the effects of statin treatment adherence and persistence on clinical outcomes reported that, in primary prevention, clinical benefits relating to CV events were observed after the first year of continued therapy. This underscores the importance of patient commitment and participation in CV disease control, and the need for further evaluation of the impact of persistent statin use on cholesterol levels and, consequently, on CV disease risk. Additionally, it should be taken into account that CV disease control must be managed by means of the joint control of all risk factors, not only through pharmacological therapy but also by lifestyles changes.21 Last, in those patients who do not achieve recommended targets, other therapeutic alternatives should be considered.22
CONCLUSIONSThe present findings demonstrate poor treatment continuity or persistence by new statin users in a population of healthy Spanish male workers. We found that a significant proportion of the study population stopped treatment soon after starting, and that the type of statin prescribed influences the likelihood of persistence. Cotreatment with antihypertensive drugs and older age were good predictors of increased persistence.
Within the context of primary prevention of CV disease, further observational drug utilization studies are required to broaden our knowledge of patterns of statin use and lead to a better understanding of the impact of individual rates of persistence on clinical outcomes. This would constitute a useful starting point to adjust prescribing recommendations when adherence and persistence rates are inadequate and, consequently, lipid targets are not achieved.
FUNDINGThe AWHS is funded by a contract signed in 2009 between the Government of Aragón (Aragon Institute of Health Sciences [IACS]) and the Health Research Fund of the Spanish Ministry of Health (National Center for Cardiovascular Research [CNIC]).This study was externally funded by a Project of the Health Research Fund, Instituto de Salud Carlos III, and the European Regional Development fund (FEDER) (PI13/01668).
CONFLICTs OF INTERESTNone declared.
- –
To provide effective treatment, statins should be taken continuously. However, treatment persistence rates for these medications are lower than expected, especially in populations with low CV risk.
- –
The causes of nonpersistence are multifactorial.
- –
This study describes poor persistence with statins in a cohort of healthy male workers.
- –
A high proportion of new statin users discontinued therapy immediately after commencing, and in numerous cases, did not recommence treatment during the following year.
- –
While some factors associated with poor persistence have been identified, there is a need for a better understanding of determinants of nonpersistence that directly affect final health outcomes.
SEE RELATED CONTENT: https://doi.org/10.1016/j.rec.2017.06.023, Rev Esp Cardiol. 2018;71:4–5.