Recovery of left ventricular ejection fraction (LVEF) has been described in alcoholic cardiomyopathy (ACM) after a period of alcohol withdrawal. Nevertheless, the prognostic impact of LVEF recovery in ACM and its determinants have not been studied. We sought to define the role of LVEF improvement in the long-term outcome of ACM and to identify predictors of LVEF recovery in these patients.
MethodsWe evaluated 101 ACM patients during a median follow-up period of 82 months [interquartile range 36–134].
ResultsAt latest follow-up, 42 patients (42%) showed substantial LVEF recovery defined as an absolute increase in LVEF ≥ 10% to a final value of ≥ 40%. Patients who recovered LVEF had better outcomes than patients who did not (heart transplant or cardiovascular death 1% vs 30%; P <.001). A QRS with <120ms (OR, 6.68; 95%CI, 2.30-19.41), beta-blocker therapy (OR, 3.01; 95%CI, 1.09-8.28), and the absence of diuretics (OR, 3.35; 95%CI, 1.08-10.42) predicted LVEF recovery in multivariate analysis. Although alcohol cessation did not predict LVEF recovery, none of the patients (n=6) who persisted with heavy alcohol consumption recovered LVEF. The rate of patients who recovered LVEF did not differ between abstainers and moderate drinkers (44% vs 45%; P=.9).
ConclusionsThe LVEF recovery is associated with an excellent prognosis in ACM. Beta-blocker treatment, QRS <120ms and absence of diuretics are independent predictors of LVEF recovery. LVEF recovery is similar in moderate drinkers and abstainers.
Keywords
Excessive alcohol use is a frequent cause of dilated cardiomyopathy (DCM), accounting for as many as 40% of cases of idiopathic DCM.1–5 Like other forms of DCM, alcoholic cardiomyopathy (ACM) is characterized by a dilated left ventricle and a reduction in the left ventricular ejection fraction (LVEF).6 Diagnosis of ACM continues to be by exclusion in patients with DCM and a long history of heavy alcohol abuse (classically >80 g of alcohol a day for at least 5 years).6,7
The injurious physical effects of alcohol have been known for centuries.8 Nonetheless, the mechanisms through which alcohol causes myocardial injury remain a focus of research.9–11 The initial event appears to be cardiomyocyte loss, either through apoptosis or necrosis; however, ACM is not distinguished from other types of DCM by histological findings (interstitial fibrosis, inflammatory infiltrates, increased fatty-acid deposition, and altered proportions of sarcomeric proteins).
Despite the clinical and epidemiological importance of ACM, only a handful of studies have investigated its natural history and associated prognostic factors.1–5,11,12 Although several series have documented LVEF recovery after a period of alcohol withdrawal, there have been no reports on how the recovered cardiac function affects the prognosis of ACM patients. Moreover, the data on whether LVEF recovery requires complete alcohol abstinence are contradictory. Several studies published almost 20 years ago identified alcohol abstinence as the main predictor of LVEF recovery in ACM1–4; however, this conclusion is questionable because, in the classic series linking LVEF recovery to alcohol withdrawal, the patient groups defined as abstainers included those who reduced their alcohol intake but maintained moderate consumption.1,2 Indeed, ACM patients who reduce their consumption to moderate levels (< 60 g/d) show similar LVEF improvements to those who withdraw from alcohol completely.13 Moreover, in our own recent study, in which most patients received current standard heart failure therapy, clinical prognosis was similar in abstainers and patients who reduced their drinking to a moderate level.12 In this earlier study on the topic,12 we identified prognostic factors associated with cardiovascular mortality and heart transplant and found that a third of ACM patients showed improvements in LVEF; however, we did not study the long-term prognostic impact of improved LVEF in ACM patients or which factors determine LVEF recovery.
Beyond describing changes in LVEF after alcohol withdrawal, no study to date has evaluated whether LVEF recovery is associated with factors other than alcohol abstinence. An ability to predict LVEF recovery in ACM would have major clinical implications, especially in relation to decisions about implanting an implantable cardioverter-defibrillator (ICD) and appropriate patient evaluation for transplant surgery.14 ICD implantation is of particular interest in ACM, since these patients have an elevated risk of ventricular arrhythmias.15
The aim of the present study was to define the long-term implications of LVEF recovery in a current series of ACM patients and to identify factors that predict LVEF recovery in this disease.
METHODSWe retrospectively analyzed prospective data collected from all consecutive ACM patients referred for evaluation to the heart failure and heart transplant units at Puerta de Hierro University Hospital in Madrid between January 1993 and December 2015. The study was approved by the hospital ethics committee and complied with the Declaration of Helsinki. The criteria for ACM diagnosis were the presence of idiopathic DCM and prolonged excessive alcohol use. Excessive alcohol consumption was defined as a self-reported alcohol intake >80g/d over a minimum period of 5 years, in line with most published ACM patient series.1–4,6,7,12 Included patients had been abusing alcohol until at least 3 months before the ACM diagnosis.
ACM patients were not enrolled in a specific alcohol recovery program, but all were recommended to cease alcohol use. Liver enzyme levels were measured during follow-up (baseline and final ALT, 83.8± 217.7 U/L and 52.7± 92.6 U/L; baseline and final AST, 27.09± 14.4 U/L and 27.69± 16.8 U/L). During follow-up, patients were classified as abstainers (complete cessation of alcohol use), moderate drinkers (consumption reduced to <80 g/d), or persistent heavy drinkers (continued consumption >80 g/d). Most patients in the moderate drinker group reduced their consumption to <20 g/d; however, the threshold for this classification was kept at 80 g/d to maintain consistency with previous studies.1–4,6,7,12
At a minimum, baseline patient evaluations included a physical examination, blood analysis, a 12-lead electrocardiogram, and echocardiography. All echocardiograms were performed at Puerta de Hierro University Hospital following standard procedures.16 Patients underwent successive invasive and prognostic investigations as needed. Successive echocardiography evaluations were not programmed at fixed intervals, but most patients had at least 1 echocardiogram a year. All patients except for 1 were examined by coronary computed tomography angiography to exclude heart disease; the exception was a 30-year-old man with no coronary risk factors and a normal treadmill stress test.
The study examined patient data from the baseline evaluation to the latest available follow-up or until death or heart transplant. All patients were followed up at regular visits to our center (at least 1 per year), and care was transferred to the referring hospital if complete LVEF recovery was stable for > 3 years. Information on patient status in December 2015 was obtained from medical records or through telephone contact with the patient or his or her referring physician.
The variables analyzed were changes in LVEF, death, and heart transplant. Cause of death was classified as heart failure, sudden cardiac death, or noncardiac cause. Significant recovery of heart function was defined as an absolute LVEF increase of ≥ 10% to a final LVEF ≥ 40%.
Statistical AnalysisCategorical variables are expressed as percentages and compared by the chi-square test and the Fisher exact test. Variables with a normal distribution are expressed as the mean±standard deviation, whereas data with a nonnormal distribution are expressed as the median [interquartile range]. Statistical comparisons were made with the Student t test, the Mann-Whitney U test, analysis of variance, and the Tukey test.
To predict significant LVEF recovery from baseline variables, we performed an initial univariate analysis of all the parameters obtained at recruitment. The criteria for including variables in the multivariate predictive model were clinical relevance and statistical significance in the univariate analysis. Alcohol cessation and variables with P <.05 in the univariate analysis were thus included in a multivariate logistic regression analysis to identify independent predictors of significant LVEF recovery.
The model was constructed by reverse selection. For the evaluation, we used the area under the curve and the Hosmer-Lemeshow test. Significance was assigned at P <.05. Data were analyzed in STAT, version 14.0 (StataCorp; College Station, Texas, United States).
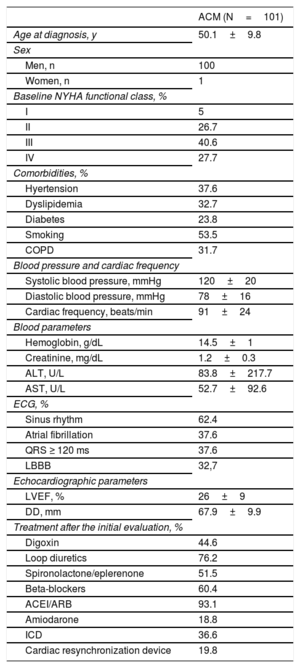
RESULTSA total of 101 ACM patients were included in the analysis (100 men; 50±10 years; LVEF, 26±9%; 68% in New York Heart Association functional class III-IV). Patients were initially evaluated during hospitalization at the center or during an outpatient visit. Study population baseline clinical characteristics, electrocardiograms, echocardiograms, and treatments are summarized in Table 1.
Clinical Characteristics and Electrocardiographic and Echocardiographic Parameters in the Baseline Evaluation of 101 Alcoholic Cardiomyopathy Patients
| ACM (N=101) | |
|---|---|
| Age at diagnosis, y | 50.1±9.8 |
| Sex | |
| Men, n | 100 |
| Women, n | 1 |
| Baseline NYHA functional class, % | |
| I | 5 |
| II | 26.7 |
| III | 40.6 |
| IV | 27.7 |
| Comorbidities, % | |
| Hyertension | 37.6 |
| Dyslipidemia | 32.7 |
| Diabetes | 23.8 |
| Smoking | 53.5 |
| COPD | 31.7 |
| Blood pressure and cardiac frequency | |
| Systolic blood pressure, mmHg | 120±20 |
| Diastolic blood pressure, mmHg | 78±16 |
| Cardiac frequency, beats/min | 91±24 |
| Blood parameters | |
| Hemoglobin, g/dL | 14.5±1 |
| Creatinine, mg/dL | 1.2±0.3 |
| ALT, U/L | 83.8±217.7 |
| AST, U/L | 52.7±92.6 |
| ECG, % | |
| Sinus rhythm | 62.4 |
| Atrial fibrillation | 37.6 |
| QRS ≥ 120 ms | 37.6 |
| LBBB | 32,7 |
| Echocardiographic parameters | |
| LVEF, % | 26±9 |
| DD, mm | 67.9±9.9 |
| Treatment after the initial evaluation, % | |
| Digoxin | 44.6 |
| Loop diuretics | 76.2 |
| Spironolactone/eplerenone | 51.5 |
| Beta-blockers | 60.4 |
| ACEI/ARB | 93.1 |
| Amiodarone | 18.8 |
| ICD | 36.6 |
| Cardiac resynchronization device | 19.8 |
ACEI, angiotensin-converting enzyme inhibitors; ACM, alcoholic cardiomyopathy; ALT, alanine aminotransferase; ARB, angiotensin II receptor blocker; AST, aspartate aminotransferase; COPD, chronic obstructive pulmonary disease; DD, diastolic diameter; ECG, electrocardiogram; ICD, implantable cardioverter-defibrillator; LBBB, left bundle branch block; LEVF, left ventricular ejection fraction; NYHA, New York Heart Association.
Unless indicated otherwise, values are expressed as mean±standard deviation.
In the baseline evaluation, all patients reported alcohol consumption >80g/d for a minimum of 5 years. During follow-up, 63% self-reported as abstainers, 31% as moderate drinkers (continued alcohol consumption but reduced to <80 g/d), and just 6% as persistent heavy drinkers, maintaining consumption above this level.
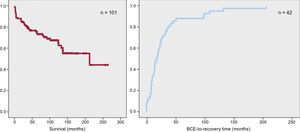
During a median follow-up of 82 months [36-134], 21 ACM patients (21%) died; 8 deaths were due to progressive heart failure, 7 to sudden cardiac death, 1 to myocardial infarction, and 5 to noncardiac causes (Figure 1). The mean follow-up duration until death was 74.5 months [3.2-137.4]. In total, 15 ACM patients (15%) received a heart transplant (3 emergency transplants and 12 elective transplants) after a mean follow-up period of 30.6 months [2.4-63.7]. A total of 6 patients (6%) had documented ventricular fibrillation at baseline. During follow-up, 37 patients received an ICD and 20 had cardiac resynchronization therapy.
A: Transplant-free survival among 101 patients with alcoholic dilated cardiomyopathy. B: Time from baseline cardiac evaluation to recovery of left ventricular ejection fraction in alcoholic cardiomyopathy patients showing significant recovery (n=42). BCE, baseline cardiac evaluation.
Data on LVEF changes were available for all patients, even though 6 patients died shortly after the baseline evaluation (< 3 months). In the latest follow-up evaluation, 42 patients (42%) showed significant heart function recovery, defined as as an absolute LVEF increase of ≥ 10% to a final LVEF ≥ 40%. Among these patients, the median interval between alcohol withdrawal and significant LVEF recovery was 28 months [9.6-30.9]; 60% of patients showing LVEF recovery passed the significance threshold within 2 years of the baseline cardiac evaluation at the center (Figure 1B).
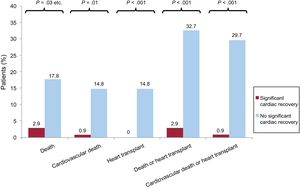
The patients who achieved significant LEVF recovery experienced better clinical progression than those who did not, having lower rates of all-cause mortality (3% vs 18%; P=.005), cardiovascular mortality (1% vs 15%; P=.002), and heart transplant (0% vs 15%; P <.0001) (Figure 2). Among ACM patients showing significant LVEF recovery, only 3 had adverse events during follow-up: 1 died of cancer, another died after a myocardial infarction, and the third had a stroke.
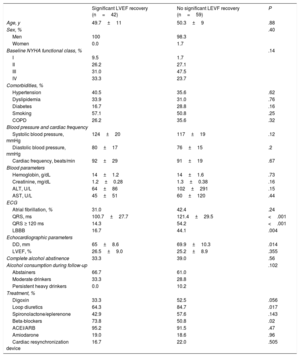
The clinical characteristics of patients with and without significant LVEF recovery are compared in Table 2, together with echocardiography and electocardiography parameters and treatments.
Echocardiographic and Electrocardiographic Parameters and Treatments in Alcoholic Cardiomyopathy Patients With or Without Left Ventricular Ejection Fraction Recovery
| Significant LVEF recovery (n=42) | No significant LEVF recovery (n=59) | P | |
|---|---|---|---|
| Age, y | 49.7±11 | 50.3±9 | .88 |
| Sex, % | .40 | ||
| Men | 100 | 98.3 | |
| Women | 0.0 | 1.7 | |
| Baseline NYHA functional class, % | .14 | ||
| I | 9.5 | 1.7 | |
| II | 26.2 | 27.1 | |
| III | 31.0 | 47.5 | |
| IV | 33.3 | 23.7 | |
| Comorbidities, % | |||
| Hypertension | 40.5 | 35.6 | .62 |
| Dyslipidemia | 33.9 | 31.0 | .76 |
| Diabetes | 16.7 | 28.8 | .16 |
| Smoking | 57.1 | 50.8 | .25 |
| COPD | 26.2 | 35.6 | .32 |
| Blood pressure and cardiac frequency | |||
| Systolic blood pressure, mmHg | 124±20 | 117±19 | .12 |
| Diastolic blood pressure, mmHg | 80±17 | 76±15 | .2 |
| Cardiac frequency, beats/min | 92±29 | 91±19 | .67 |
| Blood parameters | |||
| Hemoglobin, g/dL | 14±1.2 | 14±1.6 | .73 |
| Creatinine, mg/dL | 1.2±0.28 | 1.3±0.38 | .16 |
| ALT, U/L | 64±86 | 102±291 | .15 |
| AST, U/L | 45±51 | 60±120 | .44 |
| ECG | |||
| Atrial fibrillation, % | 31.0 | 42.4 | .24 |
| QRS, ms | 100.7±27.7 | 121.4±29.5 | <.001 |
| QRS ≥ 120 ms | 14.3 | 54.2 | <.001 |
| LBBB | 16.7 | 44.1 | .004 |
| Echocardiographic parameters | |||
| DD, mm | 65±8.6 | 69.9±10.3 | .014 |
| LVEF, % | 26.5±9.0 | 25.2±8.9 | .355 |
| Complete alcohol abstinence | 33.3 | 39.0 | .56 |
| Alcohol consumption during follow-up | .102 | ||
| Abstainers | 66.7 | 61.0 | |
| Moderate drinkers | 33.3 | 28.8 | |
| Persistent heavy drinkers | 0.0 | 10.2 | |
| Treatment, % | |||
| Digoxin | 33.3 | 52.5 | .056 |
| Loop diuretics | 64.3 | 84.7 | .017 |
| Spironolactone/eplerenone | 42.9 | 57.6 | .143 |
| Beta-blockers | 73.8 | 50.8 | .02 |
| ACEI/ARB | 95.2 | 91.5 | .47 |
| Amiodarone | 19.0 | 18.6 | .96 |
| Cardiac resynchronization device | 16.7 | 22.0 | .505 |
ACEI, angiotensin-converting enzyme inhibitors; ALT, alanine aminotransferase; ARB, angiotensin II receptor blocker; AST, aspartate aminotransferase; COPD, chronic obstructive pulmonary disease; DD, diastolic diameter; ECG, electrocardiogram; LBBB, left bundle branch block; LEVF, left ventricular ejection fraction; NYHA, New York Heart Association.
Unless indicated otherwise, values are expressed as mean±standard deviation.
The factors associated with significant LVEF recovery were the use of beta-blockers, smaller left venbtricular diastolic diameter, QRS duration <120ms, absence of left bundle branch block, and absence of diuretic therapy.
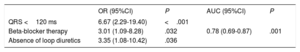
The only independent predictors of significant cardiac recovery in the multivariate regression analysis were QRS duration <120ms (odds ratio [OR]=6.68; 95% confidence interval [95%CI], 2.30-19.41), beta-blocker therapy (OR=3.01; 95%CI, 1.09-8.28), and absence of diuretics (OR=3.35; 95%CI, 1.08-10.42) (area under the curve=0.78; 95%CI, 0.69-0.87; adjusted goodness-of-fit, P=.182) (Table 3).
Independent Predictors of Left Ventricular Ejection Fraction Recovery in Alcoholic Cardiomyopathy in the Multivariate Analysis
| OR (95%CI) | P | AUC (95%CI) | P | |
|---|---|---|---|---|
| QRS <120 ms | 6.67 (2.29-19.40) | <.001 | ||
| Beta-blocker therapy | 3.01 (1.09-8.28) | .032 | 0.78 (0.69-0.87) | .001 |
| Absence of loop diuretics | 3.35 (1.08-10.42) | .036 |
AUC, area under the curve; 95%CI, 95% confidence interval; OR, odds ratio.
Variables included in the mutiple regression analysis: diastolic diameter (P=.022; 5mm diameter increments), left bundle branch block (P=.004), QRS duration <120 ms (P=.0001), complete alcohol abstinence (P=.56), beta-blocker therapy (P=.02), and absence of loop diuretics (P=.017).
None of the patients who maintained alcohol consumption >80 g/d (n=6) showed LVEF recovery. The rate of significant LVEF recovery differed markedly between persistent heavy drinkers and the combined group of ACM patients who reduced their alcohol intake to <80 g/d or abstained completely (0% vs 44%; P=.033). In contrast, the rate of LVEF recovery did not differ between moderate drinkers and abstainers (45% vs 44%; P=.9) (Figure 3).
DISCUSSIONThe present study provides the first scientific evidence of a major clinical benefit from LVEF recovery in ACM patients.
The ability to predict ACM progression has important clinical implications, especially in relation to the use of ICDs and the evaluation of patients for transplant surgery. Although several descriptive studies and patient series had reported large LVEF recoveries among ACM patients, no previous study had investigated the clinical impact of this LVEF recovery on long-term prognosis.1–3,13,17 Information on LVEF from earlier studies is mostly restricted to the description of how it changes in relation to alcohol intake during follow-up.
Our 2015 study on the natural history of ACM reported that almost a third of ACM patients showed LVEF recovery after reducing alcohol intake but did not evaluate the possible long-term prognostic implications or the determinants of this recovery.12 The present study shows that patients who experience significant cardiac recovery (absolute LVEF increase of ≥ 10% to a final LVEF ≥ 40%) have excellent clinical outcomes in long-term follow-up. ACM patients who showed significant LVEF recovery had clearly superior clinical results to those who did not. Moreover, in the present study only 1 ACM patient showing an improved LVEF died from a cardiac cause (myocardial infarction). We therefore propose that LVEF recovery to the threshold evaluated here could be used to predict clinical progression in ACM and serve as a clinical target. This conclusion is supported by our recent analysis of the incidence of malignant ventricular arrhythmias in 94 ACM patients from this same series (from the period 1993 to 2011): among ACM patients with LVEF ≥ 40%, there were no incidences of sudden cardiac death or ICD discharge.15 Identifying which ACM patients are more likely to recover LVEF past this threshold will help in decision-making about whether to implant an ICD.
Before the present study, the only factor shown to be associated with the recovery of systolic function in ACM was alcohol withdrawal.1–3,6,7 LEVF recovery has not previously been linked to any other clinical variable. Furthermore, the influence of current heart failure treatments on LVEF progression in ACM was completely unknown, since most earlier studies were conducted many years ago, before most of the currently recommended drugs and devices became available. In contrast with previous studies on ACM,1–4 most of the patients in the present study were treated for heart failure in line with current recommendations. After the baseline evaluation, more than 90% of patients were treated with angiotensin-converting enzyme inhibitor (ACEI) or angiotensin II receptor blocker (ARB) therapy, and 60% of all patients were treated with beta-blockers; for patients evaluated after 2000 (the year after the publication of the CIBIS-II study in 1999),18 the figure for beta-blockers was >80%.
Several studies have demonstrated that optimal medical treatment has a positive impact on left ventricular remodeling in unselected DCM patients and that LVEF recovery is an independent predictor of good prognosis in these patients.19,20
In the present study, a QRS duration <120ms, beta-blocker therapy, and the absence of diuretic therapy were independent predictors of significant cardiac functional recovery. These are well known prognostic factors, already identified in other studies and DCM registries.18,21,22 The beneficial effects of beta-blockers and ACEI/ARB therapy on cardiac function were demonstrated in previous studies of DCM.23,24 In the present study, ACEI/ARB therapy was not an independent predictor of LVEF recovery, probably because almost all the ACM patients (93%) received this medication from the baseline evaluation. Nevertheless, current clinical results with ACM patients12 are superior to those reported in studies from the prevasodilator era,1–4 reinforcing the conclusion that both treatments (beta-blockers and ACEI/ARB therapy) should be prescribed to all ACM patients and probably to all patients with DCM, independently of the cause. The identification of diuretic therapy as a negative prognostic indicator in the present series probably reflects the presence of congestion and more advanced heart disease in these patients at baseline. Other variables associated with fluid retention (New York Heart Association functional class III-IV and hepatojugular reflux) have not been studied in the context of LVEF recovery; however, it is interesting that these variables have been linked to worse clinical prognosis in ACM patients.4 Although atrial fibrillation has been associated with a worse prognosis, in the present study we found no significant differences in the atrial fibrillation rate between patients with LVEF recovery and those showing no significant recovery (31% vs 42.4%; P=.24).
Complete alcohol cessation has historically been considered a prerequisite for improving LVEF and achieving a better outcome in ACM.1–7,17 However, other studies have disputed the need to cease all alcohol intake in order to improve cardiac function and ACM prognosis.13,25 Moreover, while Fauchier et al. (2000) reported LVEF recocery from 27±11% to 35.4±14.6% in 28 MCA patients who stopped drinking alcohol, the same study reported that LVEF increased from 30.7±11.7% to 45.2±15% in 8 patients who continued to drink.
In contrast with that study, a study of 79 ACM and 259 DCM patients by Gavazzi et al. reported that only patients who stopped abusing alcohol showed improved LVEF.2 Interestingly, that study used a single category for abstainers and ACM patients who reduced alcohol consumption to moderate levels. Nicolás et al. retrospectively analyzed the response of LVEF to the control of alcohol consumption in an ACM patient cohort13; after a 4-year follow-up, LVEF increased not only in ACM patients who ceased all alcohol consumption, but also in patients who reduced alcohol consumption to <60g/d. Moreover, LVEF worsened only in the 8 ACM patients who maintained alcohol consumption >80 g/d. Unfortunately, that study did not include a multifactorial analysis or examine other predictors of LVEF improvement. Our present results are in line with those published by Nicolás et al.13 and conflict with those in the other studies. We found that complete alcohol abstinence was not associated with improved LVEF, either in the univariate or the multivariate analyses; rather, LVEF recovery was similar in abstainers and moderate drinkers. Also similar to the results published by Nicolás et al.,13 no significant LVEF recovery was observed in the 6 ACM patients in our study population who maintained alcohol intake above 80g/d.
Despite these results, complete alcohol abstinence is still recommended in ACM, since these patients may find it difficult to maintain moderate alcohol intake and thus run the risk of reverting to alcohol abuse, which is associated with poor LVEF development. Biological markers of alcoholism are useful in the follow-up of these patients. The main markers are mean corpuscular volume, the liver enzymes and GGT and AST, and carbohydrate-deficient transferrin, whose values increase with chronic alcohol consumption.
LimitationsDiagnosis of ACM and the classification of patients as abstainers or different subtypes of persistent drinker is based on self-reported alcohol intake, which may lead to underestimates.
The definition of ACM used here (> 80g/d of at least 5 years’ duration) is widely accepted and has been used in several previous studies; however, this cutoff probably results in an underrepresentation of women with ACM. The accepted definition of ACM does not take account of sex or body-mass index. Women are more sensitive to the effects of alcohol. Moreover, the cardiac toxicity of alcohol depends on the amount of alcohol that reaches the heart; given that women tend to have a lower body mass index, it is thus plausible that they are exposed to cardiotoxic alcohol concentrations at a lower alcohol intake.
Given that some of the patient data date back to 1993, magnetic resonance imaging data were not available for all patients. In addition, most patioents did not have an endomyocardial biopsy to exclude myocarditis; that said, none of the patients had a clinical profile indicating this condition.
Finally, the study population was obtained at a single heart transplant center, which might have biased the cohort toward patients more likely to cease or reduce alcohol consumption.
CONCLUSIONSLVEF recovery is associated with an excellent prognosis in ACM patients. Clinical variables associated with LEVF recovery were beta-blocker therapy, QRS duration <120 ms, and absence of diuretics, whereas no association was found for complete alcohol abstinence. LVEF recovery was similar in ACM patients who reduced alcohol consumption to moderate levels and those who stopped all alcohol intake. Confirmation of these results in other studies could lead to LVEF >40% being established as a clinical goal with prognostic implications.
FUNDINGThis study was partly funded by the Instituto de Salud Carlos III (grants RD12/0042/0066 and PI15/01551) with the support of the Plan Estatal de I+D+i 2013-2016: European Regional Development Fund (ERDF) “A way to make Europe”.
CONFLICTS OF INTERESTNone declared.
- –
ACM is a frequent manifestation of DCM.
- –
LVEF recovery has been described in ACM patients after a period of alcohol withdrawal.
- –
There is a lack of knowledge about the determiners and clinical impact of LVEF recovery in ACM.
- –
Significant LVEF recovery is associated with an excellent prognosis in ACM patients.
- –
Beta-blocker therapy, QRS <120 ms, and the absence of diuretic therapy are associated with LVEF recovery, but complete alcohol abstinenence is not.
- –
Achieving LVEF> 40% could be a clinical target with prognostic implications for these patients.
.
We thank Ana Royuela for help with the statistical analysis.