Catheter ablation has become the treatment of choice in an increasing number of arrhythmias in children and adolescents. There is still limited evidence of its use at a national level in Spain. The aim was to describe the characteristics and results of a modern monocentric series form a referral tertiary care centre.
MethodsRetrospective register of invasive procedures between 2004 and 2016 performed in patients under 17 years and recorded clinical characteristic, ablation methodology and acute and chronic results of the procedure.
ResultsA total of 291 procedures in 224 patients were included. Median age was 12.2 years, 60% male. Overall, 46% patients were referred from other autonomous communities. The most frequent substrates were accessory pathways (AP) (70.2%,>50% septal AP localization) and atrioventricular nodal reentrant tachycardia (AVNRT) (15.8%). Congenital and acquired heart disease was frequent (16.8%). Cryoablation was used in 35.5% of the cases. Overall acute success of the primary procedure was 93.5% (AP 93.8%; AVNRT 100%). Redo procedures after recurrence were performed in 18.9% of all substrates, with a long-term cumulative efficacy of 98.4% (AP 99.3%; AVNRT 100%). One (0.37%) serious complication occurred, a case of complete atrioventricular block.
ConclusionsOur study replicated previous international reports of high success rates with scarce complications in a high complexity series, confirming the safety and efficacy of pediatric catheter ablation in our environment performed at highly experienced referral centers.
Keywords
Catheter ablation is a safe and effective method for treating arrhythmias in the pediatric population and is considered the treatment of choice for a wide range of clinical situations and patient profiles.1 Several prospective and retrospective registries have shown a high rate of acute and long-term success and a low risk of serious complications.2–7 In addition, the increasingly widespread use of electroanatomical mapping and different ablation sources have improved safety and outcomes in complex cases. As a result, the recommendations in the clinical guidelines and expert consensus statements for performing electrophysiological procedures in adults have been extended to the pediatric population worldwide.1,8,9
However, the number of pediatric ablation procedures performed remains relatively low, and their complexity requires a high level of specialization in both pediatric cardiology and electrophysiology.8 For this reason, pediatric ablation procedures should be restricted to referral centers with adequate experience and patient volume. Compared with the extensive experience published in other parts of the world, in particular in North America, there are very few published data from Spain.
The aim of this study was to describe the characteristics and outcomes of ablation in a contemporary series of pediatric patients from a single tertiary referral center.
METHODSStudy SampleA retrospective analysis was performed of the pediatric ablation electrophysiology studies carried out at the study center between January 2004 and December 2016, by the adult electrophysiology group of the cardiology department in collaboration with pediatric cardiologists from the pediatric department. The age limit was 16 years, although second procedures performed in older patients were included. All patients were included, irrespective of their cardiac history, clinical situation, or previous ablation attempts in other centers. Data were collected on demographics such as age, sex, and region of Spain (autonomous community) from which the patients were referred, as well as weight and concomitant congenital heart disease.
Electrophysiological Study and AblationArrhythmogenic substrates were classified as atrioventricular accessory pathway (AP), atrioventricular nodal reentrant tachycardia (AVNRT), focal atrial tachycardia (FAT), macrore-entrant atrial tachycardia (MRAT), ventricular tachycardia (VT), or junctional ectopic tachycardia. The energy source (radiofrequency or cryoablation) and the approach (percutaneous or surgical) were recorded. In cases involving a change of energy source during the procedure, the last source used was recorded. Cryoablation was performed with 4, 6, and 8 mm tips (CryoCath, Medtronic Inc; Minneapolis, Minnesota,United States); when appropriate, the technique was performed with 2 applications, with cryomapping at –30°C followed by cryoablation at –70°C.
The procedures were carried out under general anesthetic. Use of 3-dimensional electroanatomic mapping systems was recorded.
Short-term and Long-term OutcomesStandard criteria were used to determine the acute success of the procedures. The criterion was the absence of conduction for 30minutes after the last application in cases of AP and was noninducibility following administration of adenosine and isoproterenol infusion for AVNRT, FAT, MRAT, and VT. For AVNRT, the presence of a nodal echo was accepted.4,8 The incidence of recurrence was recorded during protocol-based follow-up in clinic, defined as documented occurrence of the originally-treated arrhythmia and/or recurrence of pre-excitation or the corresponding electrocardiographic changes. Repeat procedures on the same substrate were recorded and the percentage of acute and long-term success was calculated for the primary procedure, as well as the long-term cumulative success of all procedures on the same substrate, defined as the absence of recurrence until the end of follow-up, excluding patients who were not scheduled for further attempts following a failed primary procedure (n=10).
Statistical AnalysisContinuous variables are reported as median [interquartile range], as they did not follow a normal distribution. They were compared using the Mann-Whitney test. Categorical variables are presented as frequency and percentage and were compared using the chi-square test or Fisher exact test. P-values < .05 were considered significant. Variables associated with acute failure or postprocedure recurrence were identified on univariate analysis and compared using multivariate logistic regression with stepwise selection or exclusion of variables. Stata 14.0 was used.
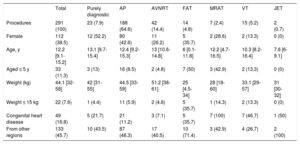
RESULTSBaseline Characteristics and IndicationsBetween January 2004 and December 2016, 291 ablation procedures were performed in 224 patients; 60% were male, and the median follow-up was 6 [2-10] years. Demographic data are shown in Table 1. The mean age was 12.2 [9.1-15.1] years; 11.4% of patients were aged ≤ 5 years and 5 patients were aged <2 years. Patients with FAT or junctional ectopic tachycardia were significantly younger than those in the other groups (median age, 6.0 and 7.6 years, respectively; P<.001). Only 4.7% of AVNRT procedures were conducted in patients aged 5 or younger. The overall median weight was 44 [32-58] kg, and 7.6% of patients met the criteria for low weight (≤ 15 kg); in FAT procedures, this rose to 35.7%. A total of 45.7% of patients were referred from regions of Spain outside the Community of Madrid.
Demographic Characteristics of the Sample According to Substrate
| Total | Purely diagnostic | AP | AVNRT | FAT | MRAT | VT | JET | |
|---|---|---|---|---|---|---|---|---|
| Procedures | 291 (100) | 23 (7.9) | 188 (64.6) | 42 (14.4) | 14 (4.8) | 7 (2.4) | 15 (5.2) | 2 (0.7) |
| Female | 112 (38.5) | 12 (52.2) | 80 (42.6) | 11 (26.2) | 5 (35.7) | 2 (28.6) | 2 (13.3) | 0 (0) |
| Age, y | 12.2 [9.1-15.2] | 13.1 [9.7-15.4] | 12.4 [9.2-15.3] | 13 [10.6-14.8] | 6 [0.1-11.8] | 12.2 [4.7-16.5] | 10.3 [8.2-16.4] | 7.6 [6-9.1] |
| Aged ≤ 5 y | 33 (11.3) | 3 (13) | 16 (8.5) | 2 (4.8) | 7 (50) | 3 (42.9) | 2 (13.3) | 0 (0) |
| Weight (kg) | 44.1 [32-58] | 42 [31-55] | 44.5 [33-59] | 51.2 [38-61] | 25 [4.5-34] | 28 [18-60] | 33.1 [29-57] | 31 [30-32] |
| Weight ≤ 15 kg | 22 (7.6) | 1 (4.4) | 11 (5.9) | 2 (4.8) | 5 (35.7) | 1 (14.3) | 2 (13.3) | 0 (0) |
| Congenital heart disease | 49 (16.8) | 5 (21.7) | 21 (11.2) | 3 (7.1) | 5 (35.7) | 7 (100) | 7 (46.7) | 1 (50) |
| From other regions | 133 (45.7) | 10 (43.5) | 87 (46.3) | 17 (40.5) | 10 (71.4) | 3 (42.9) | 4 (26.7) | 2 (100) |
AP, accessory pathway; AVNRT, atrioventricular nodal reentrant tachycardia; FAT, focal atrial tachycardia; JET, junctional ectopic tachycardia; VT, ventricular tachycardia.
Values are expressed as n (%) or median [interquartile range].
Electroanatomic navigation systems were used in 20.6% of all procedures, 15.1% with NavX (St. Jude Medical; St. Paul, Minnesota, United States) and 15.5% with CARTO (Biosense Webster; California, United States).
A total of 7.9% (n=23) of procedures were exclusively diagnostic, either due to the presence of inducible arrhythmias, anterograde and/or retrograde conduction along an AP considered low-risk, the patient or his/her parents declining the procedure after diagnosis once informed of the specific risks, and in 1 patient due to induction of unmappable polymorphic MRAT with repeated reversion to atrial fibrillation during the study. Of the therapeutic procedures, 70.2% (n=188) were performed on 1 or more APs; 15.8% (n=42) were for AVNRT; 5.2% (n=14) were for FAT; 2.6% (n=7) were for MRAT; 5.6% (n=15) for VT; and 2 cases (0.8%) were for ablation of junctional ectopic tachycardia.
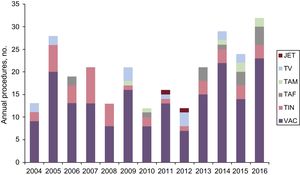
The annual distribution of the electrophysiological studies performed is shown in Figure 1. The most common indication, consistently over the years, was supraventricular tachycardia, especially AP and AVNRT. Ventricular tachycardia was a less common indication in general.
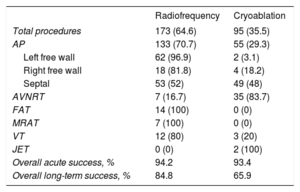
EfficacyThe distribution and outcomes of ablation according to the type of energy source used are shown in Table 2. In all, 64.6% of the procedures used radiofrequency and 35.5% used cryoablation. Choice of energy source varied according to the substrate: cryoablation was used in 83.7% of the procedures in AVNRT, in the 2 junctional ectopic tachycardias, and in 28.2% of the procedures in AP, of which 90.7% were septal. Ablation of FAT, MRAT and VT was mostly performed with radiofrequency. The acute success rate was similar for the 2 energy sources (94.2% with radiofrequency vs 93.4% with cryoablation; (P=.813); however, the recurrence rate was significantly higher with cryoablation, the overall long-term success rate being 84.8% with radiofrequency vs 65.9% with cryoablation (P=.001).
Classification and Results According to Energy Source (per Procedure)
| Radiofrequency | Cryoablation | |
|---|---|---|
| Total procedures | 173 (64.6) | 95 (35.5) |
| AP | 133 (70.7) | 55 (29.3) |
| Left free wall | 62 (96.9) | 2 (3.1) |
| Right free wall | 18 (81.8) | 4 (18.2) |
| Septal | 53 (52) | 49 (48) |
| AVNRT | 7 (16.7) | 35 (83.7) |
| FAT | 14 (100) | 0 (0) |
| MRAT | 7 (100) | 0 (0) |
| VT | 12 (80) | 3 (20) |
| JET | 0 (0) | 2 (100) |
| Overall acute success, % | 94.2 | 93.4 |
| Overall long-term success, % | 84.8 | 65.9 |
AP, accessory pathway; AVNRT, atrioventricular nodal reentrant tachycardia; FAT, focal atrial tachycardia; JET, junctional ectopic tachycardia; VT, ventricular tachycardia.
Unless otherwise indicated, values are expressed as No. (%).
Among the recurrences after a primary cryoablation procedure (which were all in patients with septal AP or AVNRT), radioablation was used for the redo procedure in only 1 patient, with success. In the remaining cases, the redo procedures used cryoablation, achieving a long-term cumulative success rate of 78%.
Irrigated tip catheters were used in 8 cases: 4 cases of AP (acute success, 75%; long-term success, 50%), 3 FAT (long-term success, 33%) and 1 VT (unsuccessful). With the exception of the FAT cases, all were redo procedures.
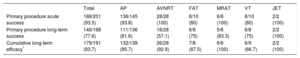
The acute and long-term overall outcomes and break-down by substrate are shown in Table 3. The acute success for primary procedures was 93.5%, including 23 procedures that had to be staged to use an irrigated tip catheter, a different energy source, or an electroanatomical navigator to achieve successful ablation. The success rate was higher in the re-entrant supraventricular tachycardias (93.8%-100%) than in FAT or VT (80%). The overall long-term success rate for primary procedures was 77.6%, although this varied according to the substrate: it was higher in AP (81.6%) than in the other substrates, particularly AVNRT (57.1%). The cumulative long-term efficacy including redo procedures on the same substrate was generally high, at 93.7%.
Acute and Long-term Outcomes According to Substrate (per Patient)
| Total | AP | AVNRT | FAT | MRAT | VT | JET | |
|---|---|---|---|---|---|---|---|
| Primary procedure acute success | 188/201 (93.5) | 136/145 (93.8) | 28/28 (100) | 8/10 (80) | 6/6 (100) | 8/10 (80) | 2/2 (100) |
| Primary procedure long-term success | 146/188 (77.6) | 111/136 (81.6) | 16/28 (57.1) | 6/8 (75) | 5/6 (83.3) | 6/8 (75) | 2/2 (100) |
| Cumulative long-term efficacy* | 179/191 (93.7) | 132/138 (95.7) | 26/28 (92.9) | 7/8 (87.5) | 6/6 (100) | 6/9 (66.7) | 2/2 (100) |
AP, accessory pathway; AVNRT, atrioventricular nodal reentrant tachycardia; FAT, focal atrial tachycardia; JET, junctional ectopic tachycardia; VT, ventricular tachycardia.
Values are expressed as no./No. (%).
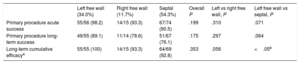
The location of the therapeutic procedures for APs is shown in Table 4. A total of 37.9% were in the left free wall (34% of procedures), 10.3% were in the right free wall (11.7% of procedures), and 51.7% were septal (54.3% of procedures). The acute success rate for primary procedures was higher in left free wall APs (98.2%) than in right free wall (93.3%) or septal (90.5%) APs. The cumulative long-term success rate was 100% for left free wall pathways, 93.3% for right free wall, and 92.8% for septal pathways, with a significant difference between left free wall and septal locations. Of the right lateral APs, 4 were Mahaim-type pathways and all had successful ablation.
Acute and Long-term Outcomes According to Location of Accessory Pathway
| Left free wall (34.0%) | Right free wall (11.7%) | Septal (54.3%) | Overall P | Left vs right free wall, P | Left free wall vs septal, P | |
|---|---|---|---|---|---|---|
| Primary procedure acute success | 55/56 (98.2) | 14/15 (93.3) | 67/74 (90.5) | .199 | .310 | .071 |
| Primary procedure long-term success | 49/55 (89.1) | 11/14 (78.6) | 51/67 (76.1) | .175 | .297 | .064 |
| Long-term cumulative efficacya | 55/55 (100) | 14/15 (93.3) | 64/69 (92.8) | .353 | .056 | <.05b |
Values are expressed as no./No. (%).
aSubstrates with a failed primary procedure that were re-treated were excluded.
Successful ablation was also performed in 5 alternating atrioventricular junctional tachycardias in the right posteroseptal area. Initially 3 procedures were performed with cryoablation, and in 1 of these there was a recurrence that was successfully treated with a second procedure without further relapse. In 2 cases, radiofrequency was used as the first option, but was unsuccessful and had to be changed to cryoablation, as the patient was on an extracorporeal membrane oxygenator for cardiogenic shock (see details below).
In 10 cases, the primary procedure was unsuccessful but redo procedures were not scheduled because the patient, family or their usual physician decided against it: 7 of these were APs, 5 of which had asymptomatic pre-excitation with no tachycardia; 2 were FAT (1 epicardial focus in the left atrial appendage and 1 in the coronary sinus ostium) in which, although they did not meet the criteria for acute success, the patients remained asymptomatic at follow-up; and there was 1 case of focal VT of the right ventricular outflow tract in which noninducibility was not achieved but which had only isolated extrasystoles at follow-up. These cases were not included in the long-term cumulative efficacy analysis.
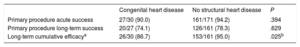
Complex CasesA total of 15.6% of patients (16.8% of the procedures) had congenital, inherited, or acquired heart disease. Within this group, 14 patients had moderate or complex congenital heart disease: single ventricle physiology (n=4), transposition of the great arteries (n=3), Ebstein anomaly (n=3), complete atrioventricular canal defect (n=2), coarctation and stenosis of the aorta (n=1), and coronary artery anomalies (n=1). The remaining patients had mild heart disease (interatrial communication [n=1], interventricular communication [n=1], multisystem disease (Kawasaki disease with coronary aneurysms [n=1], Noonan syndrome [n=1]), cardiac transplant [n=2], dilated cardiomyopathy [n=1], hypertrophic cardiomyopathy [n=2], and severe ventricular dysfunction secondary to tachycardiomyopathy [n=5]. Of this last group, 2 had cardiogenic shock and the procedure was done along with percutaneous ventricular assistance in the form of an extracorporeal membrane oxygenator, which permitted effective cryoablation following attempted radiofrequency which failed due to low blood flow.10Table 5 compares the efficacy of ablation in patients with and without congenital heart disease and shows that the acute and long-term efficacy of the primary procedures were similar, while the long-term cumulative efficacy was significantly lower in patients with congenital heart disease (86.7% vs 95.0%; P=.025).
Acute and Long-term Outcomes According to the Presence of Congenital Heart Disease
| Congenital heart disease | No structural heart disease | P | |
|---|---|---|---|
| Primary procedure acute success | 27/30 (90.0) | 161/171 (94.2) | .394 |
| Primary procedure long-term success | 20/27 (74.1) | 126/161 (78.3) | .629 |
| Long-term cumulative efficacya | 26/30 (86.7) | 153/161 (95.0) | .025b |
Values are expressed as no./No. (%).
All the patients with MRAT had congenital heart disease and all had right atrial circuits, 4 from the cavotricuspid isthmus and 3 in other locations.
Nearly half (46.7%) of the procedures for VT were performed in patients with heart disease. Most of them were due to a focal mechanism with a heterogeneous origin: 5 cases were in the right ventricle (3 in the outflow tract, 1 in the anterobasal region, 1 perihisian) and 3 in the left ventricle (1 in the outflow tract, 1 in the apex, 1 polymorphic in various regions). The remaining 2 cases were fascicular VT.
Those patients treated with a surgical approach had epicardial substrates: 1 was a right lateral AP in the patient with Kawasaki disease, treated with inferior partial sternotomy and cryoablation, and 2 had FAT originating in the left atrial appendage, treated with 3-port thoracoscopy±lateral minithoracotomy and radiofrequency. The 3 procedures were performed following a failed endocardial approach. All had long-term success.
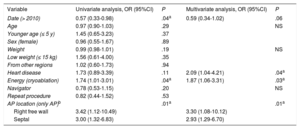
Multivariate AnalysisThe results of the multivariate analysis are shown in Table 6. The variables independently associated with procedure failure or long-term recurrence were the presence of structural heart disease (odds ratio [OR]=0.09; 95% confidence interval [95%CI], 1.04-4.21) and the use of cryoablation as the energy source (OR=1.87; 95%CI, 1.06-3.31). In the analysis of AP ablation only, a septal location (septal vs left free wall (OR=2.93; 95%CI, 1.29-6.70) or right free wall location (right free wall vs left free wall, OR=3.30; 95%CI, 1.08-10.12) was associated with worse long-term outcome. Finally, there was a tendency toward better long-term outcomes in interventions performed after 2010, but this did not reach statistical significance (OR=0.59; 95%CI, 0.34-1.02).
Predictors of Unsuccessful Acute Outcome or Long-term Recurrence
| Variable | Univariate analysis, OR (95%CI) | P | Multivariate analysis, OR (95%CI) | P |
|---|---|---|---|---|
| Date (> 2010) | 0.57 (0.33-0.98) | .04a | 0.59 (0.34-1.02) | .06 |
| Age | 0.97 (0.90-1.03) | .29 | NS | |
| Younger age (≤ 5 y) | 1.45 (0.65-3.23) | .37 | ||
| Sex (female) | 0.96 (0.55-1.67) | .89 | ||
| Weight | 0.99 (0.98-1.01) | .19 | NS | |
| Low weight (≤ 15 kg) | 1.56 (0.61-4.00) | .35 | ||
| From other regions | 1.02 (0.60-1.73) | .94 | ||
| Heart disease | 1.73 (0.89-3.39) | .11 | 2.09 (1.04-4.21) | .04a |
| Energy (cryoablation) | 1.74 (1.01-3.01) | .04a | 1.87 (1.06-3.31) | .03a |
| Navigator | 0.78 (0.53-1.15) | .20 | NS | |
| Repeat procedure | 0.82 (0.44-1.52) | .53 | ||
| AP location (only AP)b | .01a | .01a | ||
| Right free wall | 3.42 (1.12-10.49) | 3.30 (1.08-10.12) | ||
| Septal | 3.00 (1.32-6.83) | 2.93 (1.29-6.70) |
95%CI, 95% confidence interval; NS, not significant; OR, odds ratio; AP, accessory pathway.
Altogether 7 complications were recorded (2.6% of therapeutic procedures): 1 case of complete atrioventricular block (0.37% of procedures) and 6 minor complications. The complete atrioventricular block occurred in 2004 during the successful radiofrequency ablation of a fascicular VT in a heart transplant recipient. Three successful perihisian anteroseptal AP ablations developed right bundle branch block: 2 were with cryoablation and 1 with radiofrequency. One of the epicardial ablations was complicated by a small pleural effusion that resolved spontaneously. One patient with successful cryoablation of perihisian AP developed episodes of junctional ectopic tachycardia after discharge, with paroxysms of a few beats’ duration during exercise, which were self-limiting, asymptomatic, and did not require treatment. Lastly, 1 patient had partial paralysis of the hypoglossal nerve, secondary to compression from the laryngeal mask during the procedure.
DISCUSSIONIn this study, we present the results of ablation in the pediatric population in a tertiary referral center between 2004 and 2016, which show a high efficacy rate for ablation in different types of arrhythmias and substrates, with a very low complication rate. These results occurred in a highly-complex population that included patients referred from other centers, with previous failed ablations, and a high proportion of complex heart disease.
Experience in Pediatric AblationThese data are in line with previously published findings, show the current status of ablation in the pediatric population, and are comparable to those from series of adult patients.1,10,11 The current American and European consensus statements recommend catheter ablation in pediatric patients in cases of poor response to medical management or severe clinical effects (class I, level of evidence C), and as an alternative to pharmacological treatment with the aim of avoiding long-term antiarrthymic use (class I, level of evidence C).1,10 This has led to the continued increase in the number of procedures performed each year worldwide. However, the absolute number of ablations in children remains low, at around 0.05/1000 under 18-year-olds, which is approximately 1% of all ablations performed.8 The main reason for this is that these procedures are generally done in referral centers with a high patient volume and broad experience, which also focus on more complex cases. In the PACES (Pediatric and Congenital Electrophysiology Society) ablation registry, large participating centers (n=8) performed 76% of all pediatric ablation procedures.11 Other more recent European series, the Czech national registry8 and the Finnish national registry,12 published a mean annual ablation rate for referral centers of between 20 and 30 procedures per year, which is a similar number to that performed in our center. The requirements for pediatric ablation are clearly stated in the European (EHRA-AEPC, European Heart Rhythm Association-Association for European Pediatric and Congenital Cardiology) and American (PACES-HRS, Heart Rhythm Society) consensus statements.1,10 They state that procedures should be carried out by electrophysiologists with experience in pediatric ablation in collaboration with pediatric cardiologists, in centers with experience in pediatric anesthetics and pediatric cardiovascular surgery, equipped with up-to-date technology and electroanatomical navigation and cryoablation systems.1,10
There is little published evidence from Spanish series. Recently, the 2016 Spanish Catheter Ablation Registry from the Electrophysiology and Arrhythmia Working Group of the Spanish Society of Cardiology compiled, for the first time, data on the Spanish experience of pediatric ablation, which makes up 2.7% of all procedures, including details on distribution according to substrate.13 The most significant single-center registry is that by Brugada et al.,14 in which they present their initial experience with pediatric ablation, predominantly AVNRT and AP done between 1992 and 1997, with excellent results and a very low complication rate. To our knowledge, this is the first published Spanish series that includes multidisciplinary collaboration between adult and pediatric cardiologists, different access approaches and energy sources, ablation of all types of substrate, a high percentage of complex cases, and a significant number of patients with congenital heart disease, and confirms the usefulness of catheter ablation in the pediatric population performed in a tertiary referral center.
Arrhythmogenic Substrate and Ablation TechniquesRegarding patient profiles and the ablated substrate, several published single-center series have focused on supraventricular tachycardia and/or the 2 most common substrates, AP and AVNRT.4,15–17 It is also common to exclude patients with congenital heart disease, of any type or only the more complex types. This series reflects real clinical practice, with no exclusion criteria for substrate location, structural heart disease, or ablation method. If we look at the national registries and published series with no inclusion restrictions,8,12,18 we note the high percentage of complex cases in this series (> 16% of patients had congenital heart disease). In addition, there was 1 case of AP ablation that was performed with the patient on extracorporeal membrane oxygenator support, in which ventricular function normalized after the procedure.19 Another point of note is the higher prevalence of complex substrates that usually have worse outcomes, such as FAT, VT, and APs of the right free wall or septum. Lastly, although we did not manage to systematically collect data on previous ablation attempts in patients referred from other centers for a second or third ablation attempt, the high percentage of patients from other regions of the country indirectly reflects the complexity of the substrates ablated that needed a second or third procedure in our center.
This series reflects the trend over the past decade toward increased use of cryoablation as an energy source, especially for perinodal substrates, particularly AVNRT and junctional ectopic tachycardia, but also in a considerable number of septal APs. The main limitation of cryoablation is the high recurrence rate, especially in the initial published experience, but the absence of risk of complete atrioventricular block makes it the technique of choice for ablation of perinodal tachycardias in the pediatric population.15,20–25 In addition, the surgical epicardial approach, though uncommon in the pediatric population, is an option for arrhythmias with transmural or epicardial substrates or those close to the coronary arteries.
The efficacy results in this series are in line with those published in others, both single-center and prospective multicenter registries, with a high acute and long-term success rate per substrate even in patients with complex heart disease.26,27 The recurrence rate was at the upper limit of the published range, around 20%, despite–as previously discussed–the more difficult substrates, with more FAT, VT, septal APs, and cryoablation use. In general, the complications were uncommon and of little clinical relevance, with only 1 case of complete atrioventricular block, which represents 0.4% of the therapeutic procedures and was one of the complex cases from the first year of the series.24 Although described in the medical literature, there were no electrocardiographic changes compatible with ischemia after the procedure.28
LimitationsThis was a retrospective registry with all the inherent limitations of such a study design. The single-center nature limits the external validity of the results. Because of the lack of complete information on some of the procedure characteristics, we could not collect information on the use of navigation systems, the type of access to the left cavities, or mean fluoroscopy time, which would have given a more complete description of the series.
CONCLUSIONSCatheter ablation in the pediatric population can be performed with a high success rate and few complications, even in complex cases with associated heart disease, in specialized referral centers.
FUNDINGThis study was partly funded by the Cardiovascular Health Network, Instituto de Investigación Carlos III (grant numbers PI13-01882, PI13-00903, PI14/00857, PI16/01123, TEC2013-46067-R, DTS16/0160, and IJCI-2014-22178) and the ERDF (European Regional Development Fund).
CONFLICTS OF INTERESTF. Atienza is a consultant for Medtronic and Livanova.
- –
Catheter ablation is indicated for an increasing number of arrhythmias in the pediatric population. However, data are lacking on the profiles of the centers and patients involved and the safety and efficacy of the procedure.
- –
The results of this study show that ablation in pediatric patients has comparable efficacy and safety to previously published results in adults, when we included all types of arrhythmogenic substrates, ablation sources, electroanatomical navigators and highly-complex cases. These results were obtained in a tertiary referral center, in a cardiology department with a high volume of adult and pediatric patients, in collaboration with the departments of pediatrics, pediatric anesthetics, and pediatric cardiac surgery.
.